 Back
BackStructure and Stereochemistry of Alkanes
Study Guide - Smart Notes

Hydrocarbons and Alkanes
Classification of Hydrocarbons
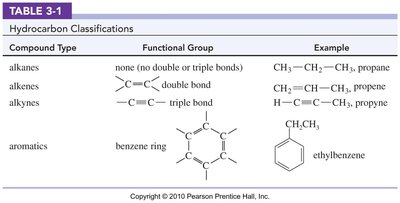
Hydrocarbons are organic molecules composed exclusively of carbon and hydrogen. They are classified based on the types of bonds and ring structures present in their molecules.
Alkanes: Saturated hydrocarbons with only single bonds. General formula: .
Alkenes: Unsaturated hydrocarbons containing at least one double bond. General formula: .
Alkynes: Unsaturated hydrocarbons containing at least one triple bond. General formula: .
Aromatics: Compounds containing a benzene ring.
Compound Type | Functional Group | Example |
|---|---|---|
alkanes | none (no double or triple bonds) | CH3CH2CH3, propane |
alkenes | C=C double bond | CH2=CHCH3, propene |
alkynes | C≡C triple bond | HC≡CCH3, propyne |
aromatics | benzene ring | ethylbenzene |
Structure and Nomenclature of Alkanes
Alkane Structures
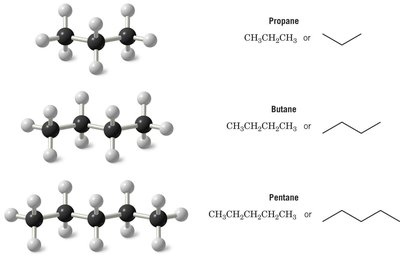
Alkanes are composed of carbon atoms connected by single bonds, with hydrogen atoms filling the remaining valencies. The simplest alkanes are methane (CH4), ethane (C2H6), propane (C3H8), butane (C4H10), and pentane (C5H12).
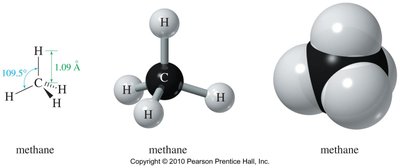
Methane: Tetrahedral geometry, bond angle 109.5°.
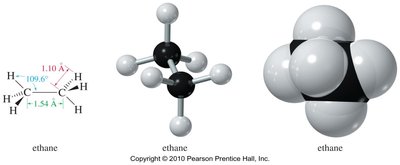
Ethane: Two tetrahedral carbons, free rotation about the C–C bond.
Propane, Butane, Pentane: Increasing chain length, more possible isomers.
IUPAC Nomenclature of Alkanes
The systematic naming of alkanes follows these steps:
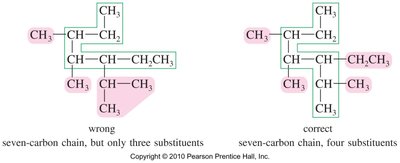
Find the longest continuous carbon chain (determines the base name).
Number the chain from the end nearest a substituent.
Name and number substituents (alkyl groups).
Alphabetize substituents, ignoring prefixes like di-, tri-, etc.
Use parentheses for complex substituents (branches on branches).
When two chains of equal length are possible, choose the one with the most substituents.
Common Alkyl Groups
Methyl: CH3–
Ethyl: CH3CH2–
Propyl: CH3CH2CH2– (n-propyl), (CH3)2CH– (isopropyl)
Butyl: CH3CH2CH2CH2– (n-butyl), sec-butyl, isobutyl, tert-butyl
Physical Properties of Alkanes
Boiling and Melting Points
Physical properties of alkanes are influenced by molecular size and branching:
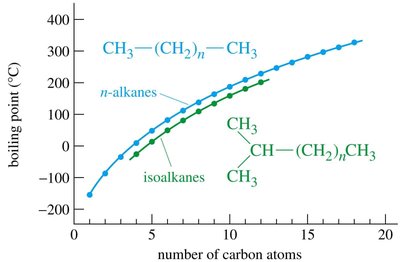
Boiling points increase with chain length (number of carbons).
Branched alkanes have lower boiling points than straight-chain isomers due to decreased surface area and weaker London dispersion forces.
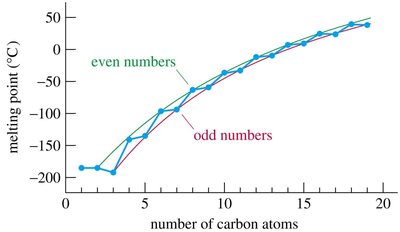
Melting points also increase with chain length, but even-numbered alkanes have higher melting points than odd-numbered ones due to better packing in the solid state.
Conformations and Stereochemistry of Alkanes
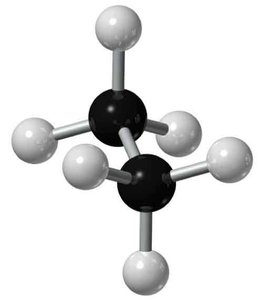
Methane and Ethane: 3D Structure
Methane and ethane are classic examples of tetrahedral geometry due to sp3 hybridization. Ethane allows free rotation about the C–C bond, leading to different conformations.
Rotation and Conformations
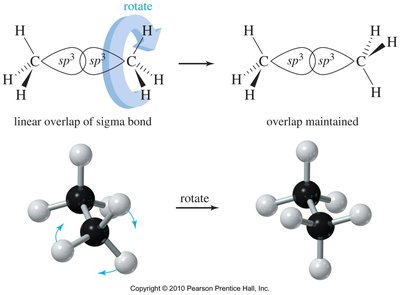
Rotation about single (sigma) bonds leads to different spatial arrangements called conformations. The most important are:
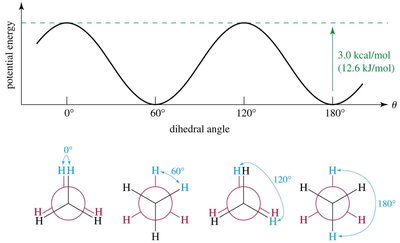
Staggered: Lowest energy, atoms as far apart as possible.
Eclipsed: Highest energy, atoms aligned with each other, leading to torsional strain.
Newman Projections
Newman projections are used to visualize conformations by looking straight down a bond axis. They are especially useful for analyzing torsional strain and steric interactions.
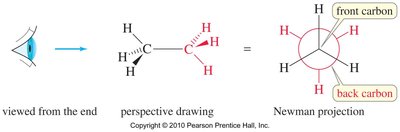
Conformational Analysis of Ethane
Ethane's staggered conformation is more stable than the eclipsed conformation by about 3.0 kcal/mol due to torsional strain.
Conformational Analysis of Propane and Butane
Propane and butane exhibit similar conformational behavior, but with increased steric interactions due to larger substituents (methyl groups).
Butane: Has two staggered conformers: anti (methyl groups 180° apart, lowest energy) and gauche (methyl groups 60° apart, higher energy).
Totally eclipsed: Highest energy, methyl groups aligned (0° dihedral angle).
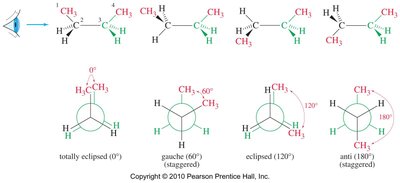
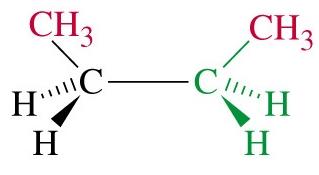
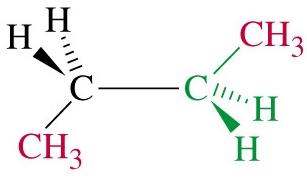
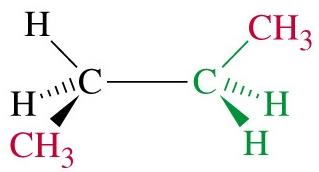
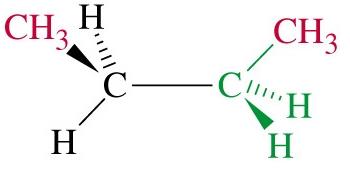
Steric Strain
Steric strain (steric hindrance) arises when bulky groups are forced close together, increasing the energy of the conformation. In butane, the totally eclipsed conformation is destabilized by steric strain between methyl groups.

Cycloalkanes and Their Stereochemistry
Structure and Properties of Cycloalkanes
Cycloalkanes are saturated hydrocarbons with carbon atoms arranged in rings. Their general formula is . The simplest is cyclopropane (C3H6).
Physical properties are similar to branched alkanes of the same carbon number.
Substituted cycloalkanes are named by numbering the ring to give substituents the lowest possible numbers.
Cis-Trans Isomerism in Cycloalkanes
Cycloalkanes with two or more substituents can exhibit cis-trans (geometric) isomerism:
Cis: Substituents on the same side of the ring.
Trans: Substituents on opposite sides of the ring.
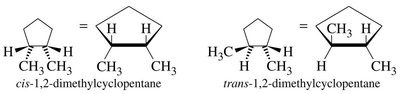
Ring Strain and Stability
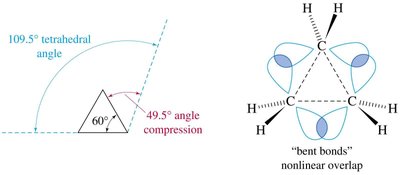
Ring strain in cycloalkanes arises from angle strain (deviation from ideal tetrahedral angle) and torsional strain (eclipsing interactions). The amount of ring strain affects the stability and reactivity of cycloalkanes.
Cyclopropane: High ring strain due to 60° bond angles and eclipsed hydrogens.
Cyclobutane: Less strain than cyclopropane, but still significant; adopts a puckered conformation to relieve torsional strain.
Cyclopentane: Minimal angle strain, puckered to reduce torsional strain.
Cyclohexane: Adopts a chair conformation with no angle or torsional strain, making it very stable.
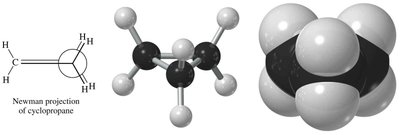
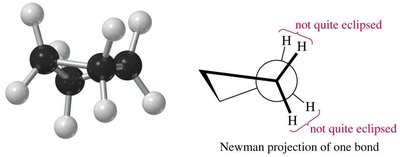
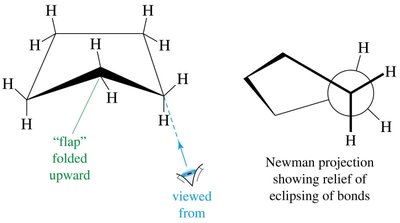
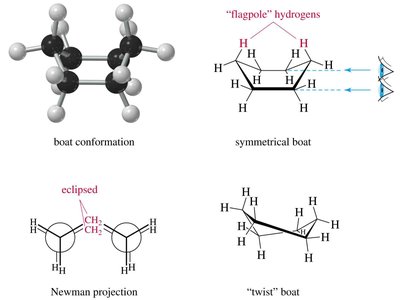
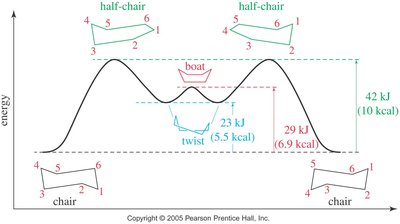
Conformations of Cyclohexane
Cyclohexane can adopt several conformations, the most important being the chair and boat forms:
Chair: All bond angles are 109.5°, all hydrogens are staggered, no ring strain.
Boat: Also has 109.5° angles but suffers from torsional strain due to eclipsed hydrogens; can twist to relieve some strain (twist-boat).
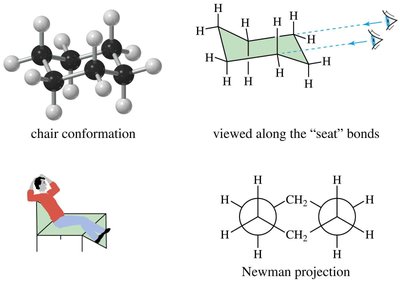
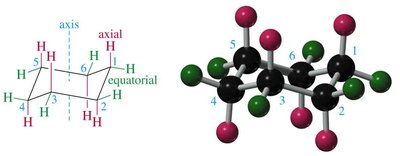
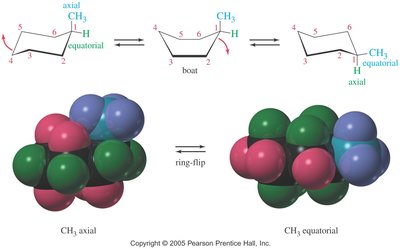
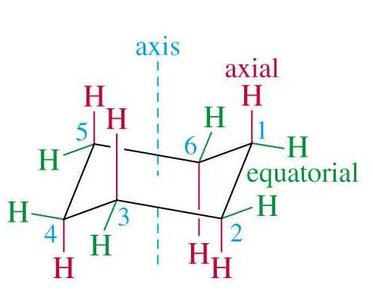
Axial and Equatorial Positions
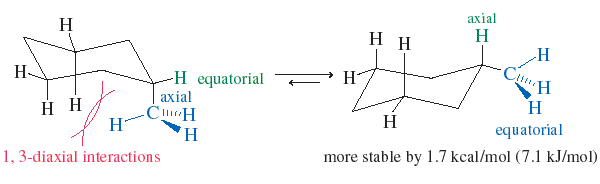
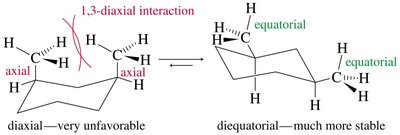
In the chair conformation, each carbon has one axial and one equatorial hydrogen. Substituents prefer the equatorial position to minimize 1,3-diaxial interactions (steric hindrance with axial hydrogens on the same side of the ring).
Disubstituted Cyclohexanes
For cyclohexanes with two substituents, the most stable conformation is usually the one with both groups equatorial. If this is not possible, the larger group will occupy the equatorial position.
Bicyclic and Polycyclic Alkanes
Bicyclic alkanes contain two fused or bridged rings. They are named using the bicyclo[x.y.z]alkane system, where x, y, and z are the number of carbons in each bridge (excluding the bridgehead carbons).
Fused rings: Share two adjacent carbons.
Bridged rings: Share two nonadjacent carbons.
Spiro compounds: Share only one carbon atom.


![Bicyclo[4.4.0]decane structure](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/bbcad7fe_image_43.png)
![Bicyclo[3.2.2]nonane structure](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/bbcad7fe_image_44.png)
Summary Table: Key Concepts in Alkane Structure and Stereochemistry
Concept | Description | Example/Note |
|---|---|---|
Alkane formula | Methane, ethane, propane | |
Isomerism | Same formula, different structure | n-butane vs isobutane |
Conformation | Spatial arrangement due to rotation | Staggered vs eclipsed ethane |
Steric strain | Repulsion between bulky groups | Totally eclipsed butane |
Ring strain | Angle/torsional strain in rings | Cyclopropane (high), cyclohexane (none) |
Axial/equatorial | Positions in cyclohexane chair | Equatorial preferred for large groups |
Bicyclic naming | bicyclo[x.y.z]alkane | bicyclo[2.2.1]heptane |
