 Back
Backch 7 Structure and Synthesis of Alkenes: Comprehensive Study Notes
Study Guide - Smart Notes

Structure and Synthesis of Alkenes
Introduction to Alkenes
Alkenes are a class of hydrocarbons characterized by the presence of at least one carbon–carbon double bond. This double bond is the functional group responsible for the unique reactivity of alkenes. Alkenes are also known as olefins, a term derived from their ability to form oily substances upon reaction.
General Formula and Hybridization
General Formula: Alkenes have the formula CnH2n, indicating two fewer hydrogens than the corresponding alkane.
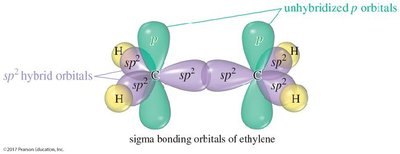
Hybridization: The carbons in the double bond are sp2 hybridized, resulting in a trigonal planar geometry with bond angles of approximately 120°.
The double bond consists of one sigma (σ) bond and one pi (π) bond. The sigma bond is formed by the overlap of sp2 orbitals, while the pi bond results from the side-by-side overlap of unhybridized p orbitals.
Bond Lengths and Angles
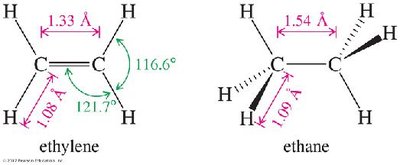
The presence of a double bond affects both bond lengths and angles in alkenes compared to alkanes:
C=C bond length: 1.33 Å (shorter than the C–C bond in alkanes, which is 1.54 Å).
Bond angles: Approximately 120° due to sp2 hybridization.
Elements of Unsaturation
Unsaturation refers to structural features that reduce the number of hydrogen atoms in a molecule compared to a saturated hydrocarbon. Each double bond or ring counts as one element of unsaturation (also called the index of hydrogen deficiency).
Calculation:
Find the number of hydrogens in the saturated formula: (2 × C) + 2
Subtract the actual number of hydrogens, then divide by 2.
Heteroatoms:
Halogens are counted as hydrogens.
Oxygen is ignored.
Nitrogen is trivalent; add the number of nitrogens to the hydrogen count.
IUPAC Nomenclature of Alkenes
Alkenes are named by identifying the longest carbon chain containing the double bond, changing the -ane suffix to -ene, and numbering the chain to give the double bond the lowest possible number.
For rings, the double bond is assumed to be between carbons 1 and 2.
Multiple double bonds are indicated with di-, tri-, or tetra- prefixes (e.g., diene, triene).
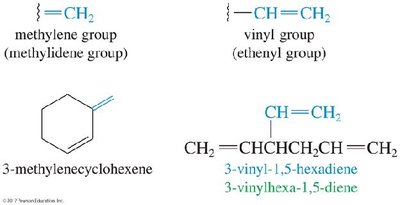
Alkenes as Substituents
When an alkene group is a substituent, it is referred to as a vinyl or methylene group, depending on its structure.
Cis-Trans (Geometric) Isomerism
Alkenes can exhibit cis-trans isomerism (geometric isomerism) when each carbon of the double bond has two different substituents. Cis isomers have similar groups on the same side, while trans isomers have them on opposite sides. Not all alkenes show this isomerism.
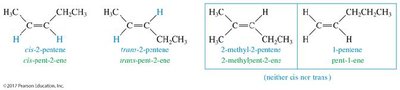
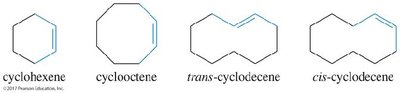
Cycloalkenes and Stereochemistry
Trans cycloalkenes are not stable unless the ring contains at least eight carbons.
All cycloalkenes are assumed to be cis unless specified otherwise.
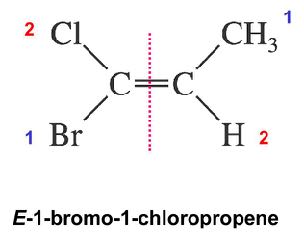
E/Z Nomenclature
The E/Z system is used for alkenes with more than two different substituents. Priorities are assigned using the Cahn–Ingold–Prelog rules:
Z (zusammen): High-priority groups on the same side.
E (entgegen): High-priority groups on opposite sides.
Stereochemistry in Dienes
For molecules with more than one double bond, the stereochemistry of each double bond must be specified (e.g., (3Z,5E)-3-bromoocta-3,5-diene).
L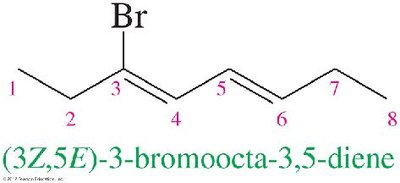
Physical Properties of Alkenes
Low boiling points, which increase with molecular mass.
Branched alkenes have lower boiling points than straight-chain isomers.
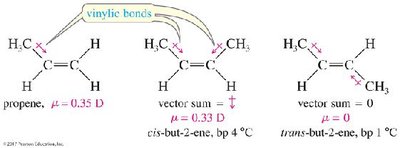
Alkenes are less dense than water and are slightly polar due to the polarizable pi bond.
Cis alkenes have higher dipole moments and boiling points than trans alkenes.
Stability of Alkenes
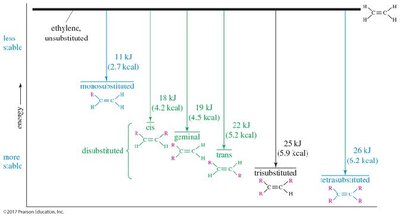
The stability of alkenes increases with the degree of substitution at the double bond. More substituted alkenes are more stable and have lower heats of hydrogenation.
Zaitsev’s Rule: In elimination reactions, the most substituted (and thus most stable) alkene is the major product.
Cycloalkenes and Ring Strain
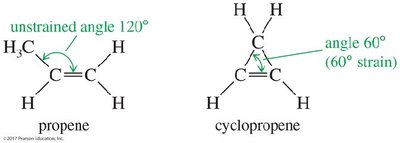
Small rings (e.g., cyclopropene) have significant ring strain, especially with double bonds.
Cis isomers are more stable than trans in small rings; trans isomers require rings of at least eight carbons to be stable.
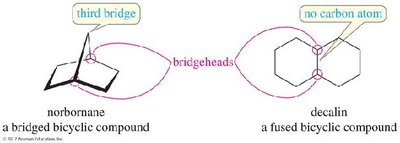
Bredt’s Rule
Bredt’s Rule states that a bridged bicyclic compound cannot have a double bond at a bridgehead position unless one of the rings contains at least eight carbon atoms. This is due to the geometric constraints that prevent proper pi bond formation in smaller rings.
Overview of Alkene Synthesis
Alkenes are commonly synthesized via elimination reactions, which remove atoms or groups from adjacent carbons to form a double bond. Major methods include:
E2 Dehydrohalogenation: Strong base, concerted mechanism.
E1 Dehydrohalogenation: Two-step mechanism via carbocation intermediate.
Dehalogenation of Vicinal Dibromides: Removal of two halogens from adjacent carbons.
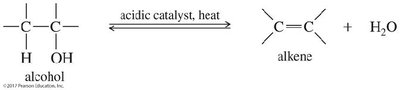
Dehydration of Alcohols: Acid-catalyzed removal of water.
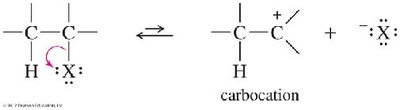
The E1 Reaction Mechanism
The E1 (unimolecular elimination) reaction proceeds in two steps:
Ionization to form a carbocation intermediate (rate-determining step).
Deprotonation by a base to form the alkene.
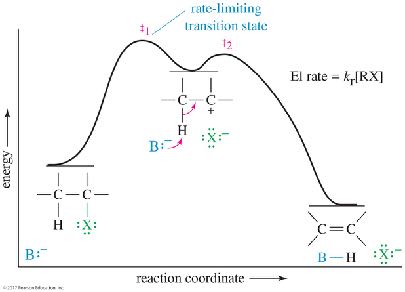
E1 Energy Diagram
The rate-determining step is the formation of the carbocation. The energy profile is similar to that of the SN1 reaction, as both share the same first step.
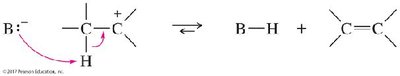
The E2 Reaction Mechanism
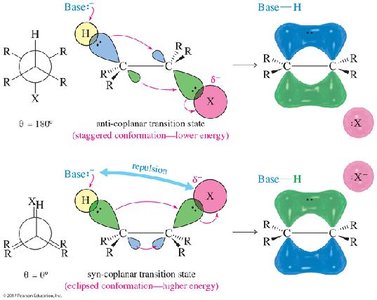
The E2 (bimolecular elimination) reaction is a one-step, concerted process requiring a strong base. The proton is abstracted, the double bond forms, and the leaving group departs simultaneously. The reaction is stereospecific, requiring the hydrogen and leaving group to be anti-coplanar (180° apart).
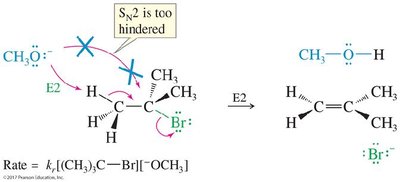
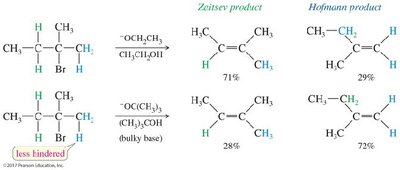
Zaitsev and Hofmann Products
When a bulky base is used in E2 eliminations, the less substituted (Hofmann) product may predominate due to steric hindrance, contrary to Zaitsev’s rule.
E2 Stereochemistry
The E2 reaction requires an anti-coplanar arrangement of the hydrogen and leaving group for optimal orbital overlap and minimal steric hindrance. This requirement explains the regio- and stereoselectivity observed in E2 eliminations.
Substitution vs. Elimination
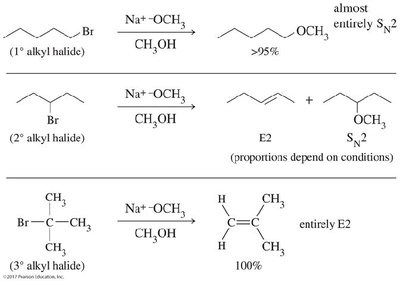
The competition between substitution and elimination depends on several factors:
Primary halides: Favor SN2 with strong nucleophiles.
Secondary halides: Can undergo SN2, E2, SN1, or E1 depending on nucleophile strength and reaction conditions.
Tertiary halides: Favor E2 with strong bases, SN1/E1 with weak nucleophiles.
High temperature and bulky bases favor elimination.
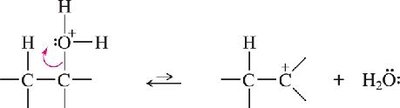
Dehydration of Alcohols
Alcohols can be dehydrated to form alkenes using concentrated acids (H2SO4 or H3PO4) and heat. The reaction proceeds via an E1 mechanism, often with rearrangements, and follows Zaitsev’s rule.

Summary Table: Key Properties and Reactions of Alkenes
Property/Reaction | Description |
|---|---|
Hybridization | sp2 (trigonal planar, 120° angles) |
Bond Length | C=C: 1.33 Å; C–C (alkane): 1.54 Å |
Isomerism | Cis-trans (geometric), E/Z nomenclature |
Physical Properties | Low boiling point, slightly polar, less dense than water |
Stability | Increases with substitution; Zaitsev’s rule applies |
Synthesis | E1, E2, dehalogenation, dehydration |
Reactivity | Electrophilic addition, polymerization, oxidation |
