 Back
BackThe Chemistry of Nucleic Acids: Structure, Function, and Mechanisms
Study Guide - Smart Notes

Chapter 26: The Chemistry of the Nucleic Acids
Introduction to Nucleic Acids
Nucleic acids, including DNA and RNA, are essential biomolecules responsible for the storage, transmission, and expression of genetic information. Their structure and chemistry underpin the molecular mechanisms of heredity and protein synthesis.
Structure and Components of Nucleic Acids
Phosphodiester Backbone
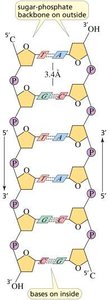
The backbone of nucleic acids consists of alternating sugar and phosphate groups linked by phosphodiester bonds. These bonds connect the 3'-hydroxyl group of one sugar to the 5'-phosphate group of the next, forming a stable, directional polymer.
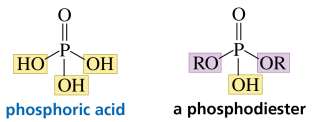
Phosphoric acid forms the basis for the phosphate group in nucleic acids.
Phosphodiester bonds are crucial for the integrity and polarity of nucleic acid strands.
Nitrogenous Bases
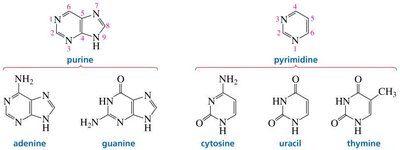
Nucleic acids contain two types of nitrogenous bases: purines and pyrimidines. These bases are responsible for the encoding of genetic information and the specificity of base pairing.
Purines: Adenine (A) and Guanine (G)
Pyrimidines: Cytosine (C), Thymine (T, found only in DNA), and Uracil (U, found only in RNA)
Nucleosides and Nucleotides
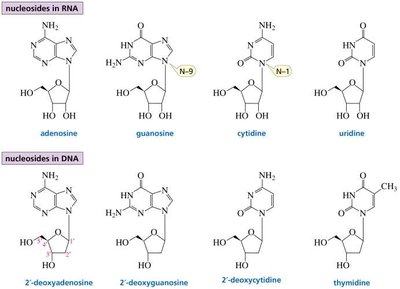
A nucleoside consists of a nitrogenous base linked to a pentose sugar (ribose in RNA, deoxyribose in DNA). A nucleotide is a nucleoside with one or more phosphate groups attached to the 5' carbon of the sugar.
Nucleosides in RNA: Adenosine, Guanosine, Cytidine, Uridine
Nucleosides in DNA: 2'-Deoxyadenosine, 2'-Deoxyguanosine, 2'-Deoxycytidine, Thymidine
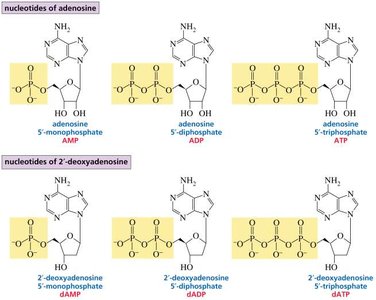
Nucleotides: Addition of phosphate groups forms nucleotides such as AMP, ADP, ATP (adenosine mono-, di-, and triphosphate), and their deoxy analogs in DNA.
Nomenclature of Nucleotides
The nomenclature distinguishes between ribonucleotides (RNA) and deoxyribonucleotides (DNA), as well as the specific base present. For example, adenosine 5'-phosphate (AMP) vs. 2'-deoxyadenosine 5'-phosphate (dAMP).
Thymine is found only in DNA; Uracil is found only in RNA.
Cyclic Nucleotides
Cyclic AMP (cAMP) is a nucleotide derivative that acts as a second messenger in cellular signaling, linking hormone action to cellular responses.
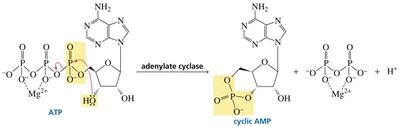
Biosynthesis and Structure of Nucleic Acids
Polymerization of Nucleotides
Nucleic acids are polymers of nucleotides. The polymerization process involves the formation of phosphodiester bonds, with the release and hydrolysis of pyrophosphate making the reaction energetically favorable and irreversible.
Dinucleotide: Two nucleotide subunits
Oligonucleotide: 3–10 nucleotide subunits
Polynucleotide: Many nucleotide subunits
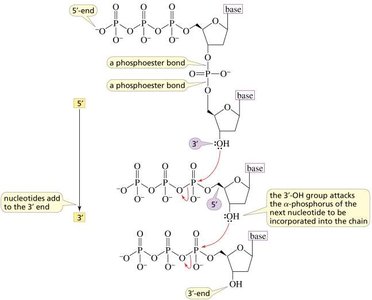
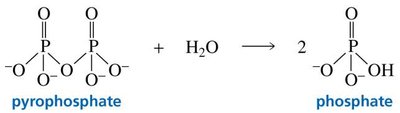
Primary Structure of Nucleic Acids
The primary structure is the linear sequence of nucleotides, written from the 5' to the 3' end. This sequence encodes genetic information.
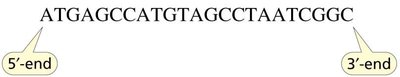
DNA Structure and Base Pairing
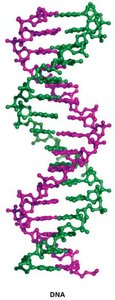
Double-Stranded DNA and the Double Helix
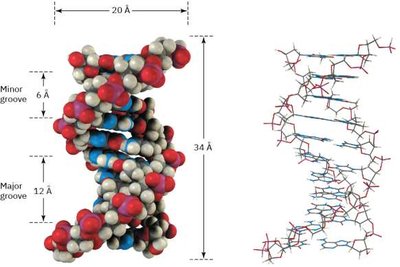
DNA is typically double-stranded, with two antiparallel strands wound into a right-handed double helix. The sugar-phosphate backbone is on the outside, and the bases are on the inside, paired by hydrogen bonds.
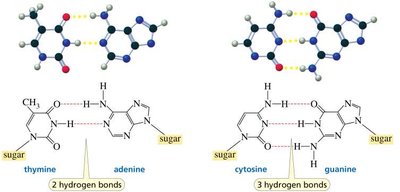
Complementary base pairing: Adenine pairs with Thymine (A–T) via two hydrogen bonds; Guanine pairs with Cytosine (G–C) via three hydrogen bonds.
The Double Helix and Grooves
The double helix has major and minor grooves, which are important for protein-DNA interactions and the binding of regulatory molecules.
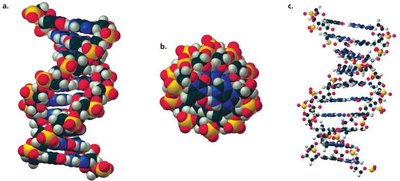
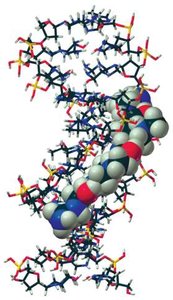
Antibiotic Binding and DNA Stability
Certain antibiotics bind in the minor groove of DNA, interfering with replication and transcription, which is a basis for their biological activity.
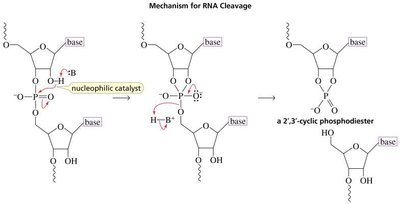
Stability: DNA vs. RNA
DNA lacks a 2'-hydroxyl group, making it more chemically stable than RNA. The 2'-OH in RNA acts as a nucleophilic catalyst, promoting cleavage and resulting in a much faster rate of hydrolysis compared to DNA.
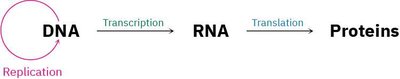
Genetic Information Flow: Replication, Transcription, and Translation
Central Dogma of Molecular Biology
The central dogma describes the flow of genetic information: DNA is replicated, transcribed into RNA, and translated into proteins.
DNA Replication
During replication, each DNA strand serves as a template for the synthesis of a complementary strand. The process is semi-conservative, and synthesis occurs in the 5' to 3' direction.
The leading strand is synthesized continuously, while the lagging strand is synthesized discontinuously in short fragments.
RNA Transcription
Transcription is the synthesis of RNA from a DNA template. The RNA sequence is complementary to the DNA template strand, with uracil (U) replacing thymine (T).
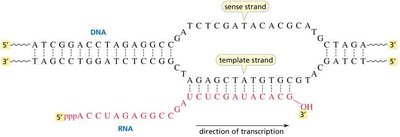
Types of RNA
mRNA (messenger RNA): Encodes the sequence of amino acids in a protein.
rRNA (ribosomal RNA): Structural and catalytic component of ribosomes.
tRNA (transfer RNA): Carries amino acids to the ribosome for protein synthesis.
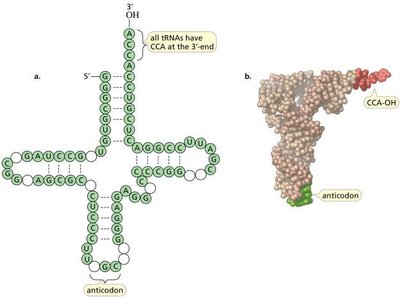
tRNA Charging and Aminoacyl-tRNA Synthetases
Amino acids are attached to their corresponding tRNAs by aminoacyl-tRNA synthetases, ensuring the correct amino acid is incorporated during translation. The enzyme has proofreading ability to correct errors in amino acid attachment.
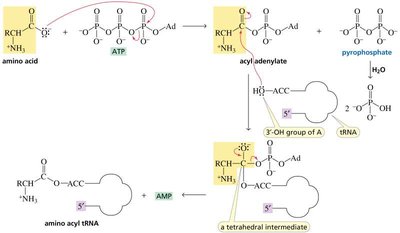
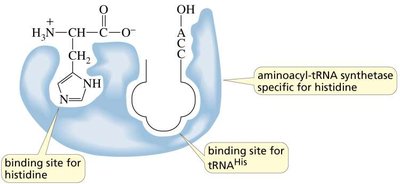
The Genetic Code and Protein Synthesis
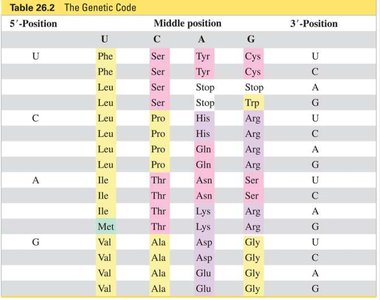
The Genetic Code
The genetic code is a set of triplet codons in mRNA that specify amino acids. It is nearly universal and degenerate, meaning multiple codons can code for the same amino acid.
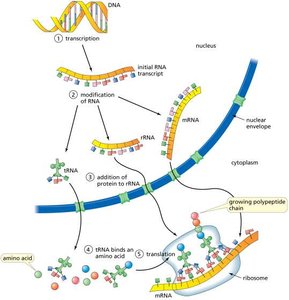
Translation
Translation is the process by which ribosomes synthesize proteins using mRNA as a template. tRNAs bring amino acids to the ribosome, matching their anticodon to the codon on the mRNA.
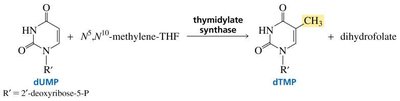
Special Topics in Nucleic Acid Chemistry
Methylation and Thymine Formation
Thymine in DNA is formed by methylation of deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP), a reaction catalyzed by thymidylate synthase and requiring tetrahydrofolate as a cofactor.
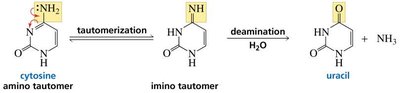
Why DNA Contains Thymine Instead of Uracil
DNA uses thymine instead of uracil to allow for the detection and repair of cytosine deamination, which would otherwise result in uracil and potential mutations.
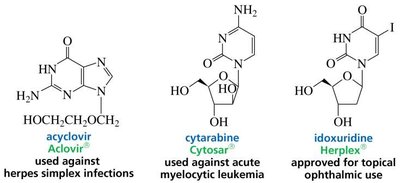
Antiviral Drugs
Many antiviral drugs act by interfering with viral nucleic acid synthesis, often by mimicking nucleoside or nucleotide structures and inhibiting viral polymerases.
Restriction Enzymes
Restriction enzymes (endonucleases) recognize specific palindromic DNA sequences and cleave the DNA at these sites, a key tool in molecular biology and genetic engineering.

DNA Sequencing
DNA sequencing involves the synthesis of new DNA strands using protected nucleoside triphosphates and DNA polymerase, allowing the determination of nucleotide order.
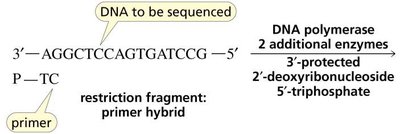
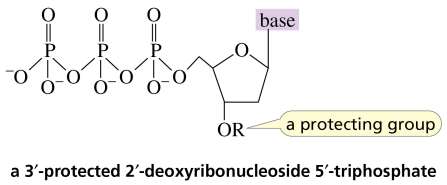
Genetic Engineering Example
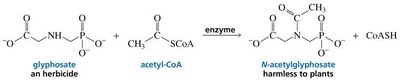
Genetic engineering can introduce enzymes into plants that inactivate herbicides, conferring resistance and allowing selective weed control.
Summary Table: Nucleic Acid Components
Base | Ribonucleoside | Deoxyribonucleoside | Ribonucleotide | Deoxyribonucleotide |
|---|---|---|---|---|
Adenine | Adenosine | 2'-Deoxyadenosine | Adenosine 5'-phosphate | 2'-Deoxyadenosine 5'-phosphate |
Guanine | Guanosine | 2'-Deoxyguanosine | Guanosine 5'-phosphate | 2'-Deoxyguanosine 5'-phosphate |
Cytosine | Cytidine | 2'-Deoxycytidine | Cytidine 5'-phosphate | 2'-Deoxycytidine 5'-phosphate |
Thymine | — | Thymidine | — | Thymidine 5'-phosphate |
Uracil | Uridine | — | Uridine 5'-phosphate | — |
Key Equations
Hydrolysis of pyrophosphate:
Base pairing (hydrogen bonds):
A–T: 2 hydrogen bonds
G–C: 3 hydrogen bonds
Learning Objectives
Describe the difference between a nucleoside and a nucleotide.
Name the five nucleotides.
Describe the mechanism for the formation of nucleic acids from nucleotide triphosphates.
Explain why DNA does not have a 2'-OH group.
Explain why the bases in DNA are A, G, C, and T, whereas the bases in RNA are A, G, C, and U.
Describe replication, transcription, and translation.
