15. Chemical Kinetics
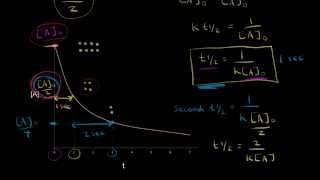
Half-Life

Get help from an AI Tutor
Ask a question to get started.
Problem 99b
Textbook Question
Textbook QuestionAmericium-241 is used in smoke detectors. It has a first-order rate constant for radioactive decay of k = 1.6 * 10-3 yr-1. By contrast, iodine-125, which is used to test for thyroid functioning, has a rate constant for radioactive decay of k = 0.011 day-1. (c) How much of a 1.00-mg sample of each isotope remains after three half-lives?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
787
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos