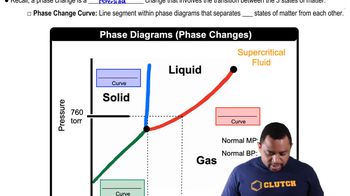
The phase diagram for neon is
Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point?

 Verified step by step guidance
Verified step by step guidance

The phase diagram for neon is
Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point?
Use the phase diagram of neon to answer the following questions. (a) What is the approximate value of the normal boiling point?
Use the phase diagram of neon to answer the following questions. (b) What can you say about the strength of the intermolecular forces in neon and argon based on the critical points of Ne and Ar (see Table 11.5.)?
Indicate whether each statement is true or false: (c) Molecules that exhibit a liquid crystalline phase do so at well-defined temperatures and pressures.
In all four liquid crystalline phases shown in Figure 11.32, the long axis of the molecule preferentially orders along one or more specific directions. In three of the four phases the molecules also lose some freedom of translational motion. In which of the four liquid crystalline phases do the molecules retain the freedom to move in all three directions that they possess in the liquid phase: nematic, smectic A, smectic C, or cholesteric?