 Back
BackDNA Structure: Nucleotides, Double Helix, and Chemical Composition
Study Guide - Smart Notes

DNA Structure
Nucleotides: The Building Blocks of Nucleic Acids
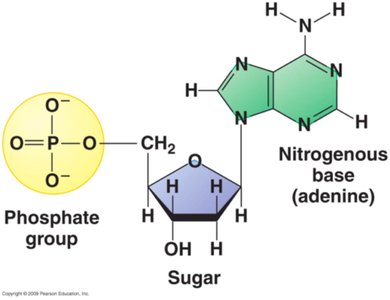
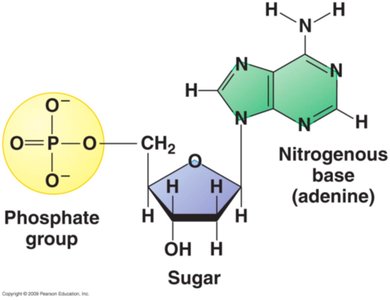
Nucleotides are the fundamental units that make up DNA and RNA, the nucleic acids responsible for genetic information storage and transmission. Each nucleotide consists of three components: a pentose (5-carbon) sugar, a nitrogenous base, and a phosphate group.
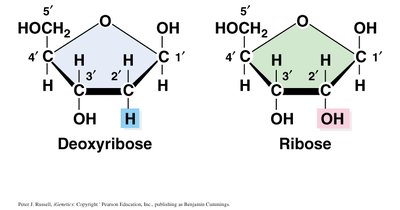
Pentose Sugar: DNA contains deoxyribose, while RNA contains ribose. The difference lies at the 2' carbon: deoxyribose has a hydrogen atom, ribose has a hydroxyl group.
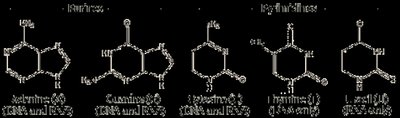
Nitrogenous Base: Bases are classified as purines (adenine, guanine) and pyrimidines (cytosine, thymine in DNA; uracil in RNA).
Phosphate Group: Attached to the 5' carbon of the sugar, it links nucleotides together via phosphodiester bonds.
Nucleosides and Nucleotides
A nucleoside consists of a nitrogenous base and a pentose sugar. When a phosphate group is added, it becomes a nucleotide. Nucleotides are the monomers that polymerize to form nucleic acids.
Nucleoside: Base + Sugar
Nucleotide: Nucleoside + Phosphate group
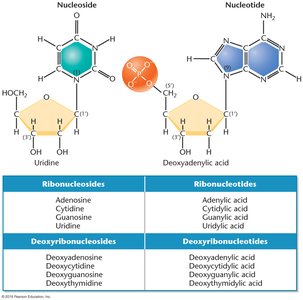
Chemical Composition of DNA and RNA
DNA and RNA differ in both their sugar and nitrogenous base composition:
DNA: Contains deoxyribose sugar and the bases adenine (A), cytosine (C), guanine (G), and thymine (T).
RNA: Contains ribose sugar and the bases adenine (A), cytosine (C), guanine (G), and uracil (U) (uracil replaces thymine).
Polynucleotide Chains and Phosphodiester Bonds
Formation and Stability of DNA Strands
Nucleotides are joined together by phosphodiester bonds, which are covalent bonds between the phosphate group at the 5' carbon of one nucleotide and the 3' carbon of the sugar of another. This linkage forms the backbone of DNA and RNA, providing remarkable stability to the molecule.
Polynucleotide Chain: A sequence of nucleotides linked by phosphodiester bonds.
Polarity: DNA strands have directionality, with a 5' end (phosphate group) and a 3' end (hydroxyl group).
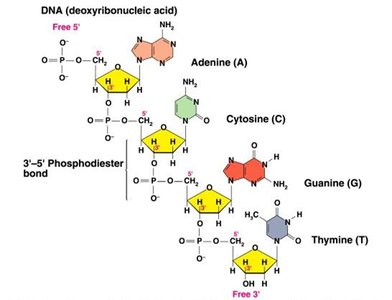
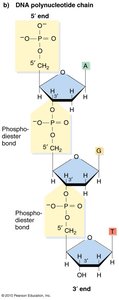
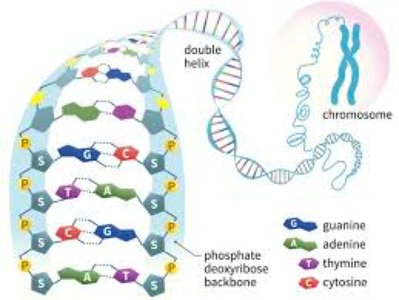
Base Pairing and Double Helix Structure
Complementary Base Pairing
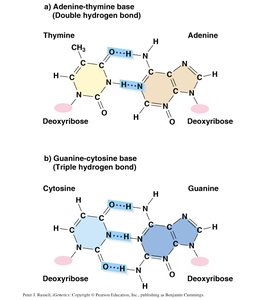
DNA consists of two antiparallel strands held together by hydrogen bonds between complementary bases. Each base pair consists of a purine and a pyrimidine:
Adenine (A) pairs with Thymine (T) via two hydrogen bonds.
Guanine (G) pairs with Cytosine (C) via three hydrogen bonds (making G-C pairs more stable).
Watson & Crick Double Helix Model
The double helix model of DNA, proposed by Watson and Crick in 1953, describes the structure as two polynucleotide chains wound in a right-handed helix. The sugar-phosphate backbones are on the outside, and the bases are oriented toward the central axis.
Antiparallel Strands: One strand runs 5' to 3', the other 3' to 5'.
Major and Minor Grooves: The backbones are not equally spaced, creating grooves that are important for protein-DNA interactions.
Helical Parameters: Base pairs are 0.34 nm apart; one complete turn of the helix is 3.4 nm (10 base pairs per turn).
Chargaff's Rules and Base Composition
Erwin Chargaff's studies revealed that the amount of adenine equals thymine, and the amount of guanine equals cytosine in DNA. The overall %GC or %AT content varies among organisms.
Organism | %A | %T | %G | %C |
|---|---|---|---|---|
Homo sapiens | 31.0 | 31.5 | 19.1 | 18.4 |
Drosophila | 27.3 | 27.6 | 22.5 | 22.5 |
Aythya americana | 25.8 | 25.8 | 24.2 | 24.2 |
X-ray Diffraction and Helical Structure
Rosalind Franklin and Maurice Wilkins used X-ray diffraction to show that DNA is a helical structure with distinctive spacing: 0.34 nm between base pairs and 3.4 nm per helical turn.
Summary of Key Features of DNA Double Helix
Two polynucleotide chains wound in a right-handed double helix
Antiparallel orientation of strands
Sugar-phosphate backbone on the outside, bases on the inside
Complementary base pairing via hydrogen bonds
Major and minor grooves due to unequal backbone spacing
Helical parameters: 0.34 nm per base pair, 3.4 nm per turn
Key Equations
Phosphodiester Bond Formation:
Chargaff's Rule:
