 Back
BackGenes, Proteins, Enzymes, and Human Disease: Molecular Genetics Study Guide
Study Guide - Smart Notes

Genes and Their Role in Human Disease
Historical Perspective: Inborn Errors of Metabolism
Mutations in genes can cause human diseases, as first described by Archibald Garrod in 1902. He studied alkaptonuria, a disease characterized by black urine and arthritis, which results from the accumulation of homogentisic acid. Garrod proposed that this was an 'inborn error of metabolism,' not caused by germs, and suggested it was a recessive Mendelian trait. His work implied that genes are required for specific chemical reactions in the body.

Key Point: Alkaptonuria is caused by a defective enzyme in the pathway for breaking down phenylalanine, leading to the accumulation of homogentisic acid.
What is a Gene?
Genetic and Biochemical Definitions
Genes have been defined genetically as units controlling aspects of phenotype, residing on chromosomes and segregating in defined ways. Biochemically, a gene is a segment of DNA containing information to express a protein that performs a function in the cell or body.
Proteins: Structure and Function
Amino Acids and Protein Structure
Proteins are polymers made of amino acids, each with a unique sequence. There are 20 common amino acids, grouped by their R side chains into categories: nonpolar, polar, acidic, and basic. The sequence and chemical properties of amino acids determine protein folding and function.
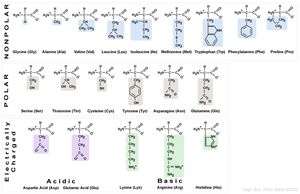
Key Point: The chemical nature of the R group determines whether an amino acid is hydrophilic (polar/charged, found on the outside of proteins) or hydrophobic (nonpolar, found on the inside).
Peptide Bonds and Protein Chains
Amino acids are joined by peptide bonds, forming linear chains. Polypeptide chains have a free amino group at the start (amino terminus) and a free carboxyl group at the end (carboxyl terminus).
Levels of Protein Structure
Proteins fold into precise 3D structures based on their amino acid sequence. There are four levels of protein structure:
Primary structure: Linear sequence of amino acids joined by covalent bonds.
Secondary structure: Local structures formed by hydrogen bonds (e.g., alpha helix, beta sheet).
Tertiary structure: Final 3D shape formed by long-range interactions (hydrogen bonds, ionic bonds, hydrophobic/hydrophilic effects).
Quaternary structure: Multiple polypeptides working together (e.g., hemoglobin has 2 alpha and 2 beta subunits).
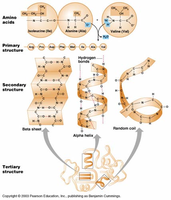

Example: Hemoglobin is composed of four polypeptide subunits, demonstrating quaternary structure.
Genes, Proteins, and Enzymes
Enzymes as Proteins
Most genes encode proteins, many of which are enzymes. Enzymes are proteins that catalyze specific chemical reactions, folding to create an active site where the reaction occurs.
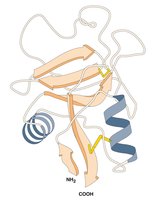
Key Point: Enzymes perform chemical reactions essential for metabolism; mutations in genes encoding enzymes can disrupt these pathways and cause disease.
Biochemical Pathways and Genetic Diseases
Alkaptonuria and Phenylketonuria (PKU)
Alkaptonuria is caused by a defective enzyme in the phenylalanine breakdown pathway, leading to homogentisic acid accumulation. Phenylketonuria (PKU) results from mutations in the gene encoding phenylalanine hydroxylase (PAH), causing phenylalanine to accumulate and produce harmful byproducts.
Example: PKU is screened in newborns, and affected children are placed on a low-phenylalanine diet to prevent neurological damage.
Tay-Sachs Disease
Tay-Sachs disease is caused by a defect in the lysosomal enzyme HEXA, leading to substrate accumulation and neuronal degeneration. It is common among individuals of Jewish ancestry and has no cure, but genetic testing has reduced its prevalence.
Sickle Cell Anemia: A Molecular Disease
Genetic Basis and Molecular Change
Sickle cell anemia is caused by mutations in the gene for beta hemoglobin. Hemoglobin is the oxygen-carrying molecule in red blood cells, composed of two alpha and two beta chains. A single amino acid change (E6V: glutamic acid to valine at position 6) alters the protein's structure, causing red blood cells to adopt a sickle shape under low oxygen conditions.
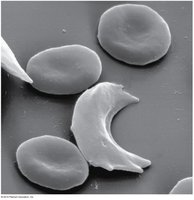

Key Point: The E6V mutation leads to hemoglobin aggregation, sickling of red blood cells, and symptoms such as pain, weakness, and increased risk of infection.
Protein Gel Electrophoresis
Linus Pauling demonstrated that hemoglobin from normal and affected individuals behaves differently in gel electrophoresis, identifying sickle cell anemia as a molecular disease.
Population Genetics: Sickle Cell and Malaria
Sickle cell anemia is prevalent in populations exposed to malaria. Heterozygous carriers (bAbS) have increased fitness in malarial environments because their red blood cells have a shorter lifespan, disrupting the malaria parasite's life cycle. This explains the persistence of the sickle cell allele in these populations.
Example: The sickle cell allele is common in African, Caribbean, and Brazilian populations due to historical exposure to malaria.
Beadle and Tatum: One Gene-One Enzyme Hypothesis
Model Organisms and Genetic Screens
Beadle and Tatum used the bread mold Neurospora crassa to show that one gene controls one enzyme at a specific step in a biochemical pathway. They exposed spores to mutagens, isolated auxotrophic mutants, and determined which compounds the mutants could not synthesize.
Key Point: Their work led to the 'one gene-one enzyme' hypothesis, later refined to 'one gene-one polypeptide.'
Summary Table: Amino Acid Categories
The 20 amino acids are grouped by their chemical properties:
Category | Amino Acids |
|---|---|
Nonpolar | Glycine, Alanine, Valine, Leucine, Isoleucine, Methionine, Tryptophan, Phenylalanine, Proline |
Polar | Serine, Threonine, Cysteine, Tyrosine, Asparagine, Glutamine |
Acidic | Aspartic acid, Glutamic acid |
Basic | Lysine, Arginine, Histidine |

Key Equations and Concepts
Peptide bond formation:
Central Dogma:
Mutation effect:
Additional info: The notes cover Ch. 4 (Gene Interaction), Ch. 10 (Eukaryotic Chromosome Abnormalities and Molecular Organization), and related sections on protein structure, gene function, and genetic diseases, making them highly relevant to a college genetics course.
