 Back
BackGenes, Proteins, Enzymes, and Human Disease: Molecular Genetics Study Guide
Study Guide - Smart Notes

Genes and Their Role in Human Disease
Historical Perspective: Inborn Errors of Metabolism
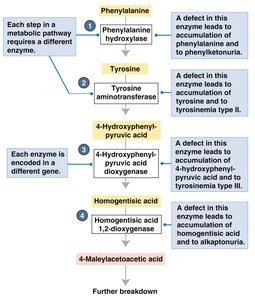
Mutations in genes can cause human diseases, as first described by Archibald Garrod in 1902. He studied alkaptonuria, a disease characterized by black urine and arthritis, which results from the accumulation of homogentisic acid. Garrod proposed that this was an 'inborn error of metabolism,' not caused by germs, and suggested it was a recessive Mendelian trait.
Key Point: Alkaptonuria is caused by a defective enzyme in the pathway breaking down phenylalanine.
Key Point: Genes are required for specific chemical reactions in the body.
Example: Alkaptonuria demonstrates how a single gene mutation can disrupt a metabolic pathway.

What is a Gene?
Genetic and Biochemical Definitions
Genes have been defined both genetically and biochemically. Genetically, a gene controls some aspect of an organism’s phenotype and resides on chromosomes, segregating in defined ways. Biochemically, a gene is a segment of DNA containing information to express a protein that performs a function in the cell or body.
Key Point: Genes encode proteins, which are responsible for cellular functions.
Key Point: Mutations in genes can alter protein function, leading to disease.
Proteins: Structure and Function
Amino Acids and Protein Structure
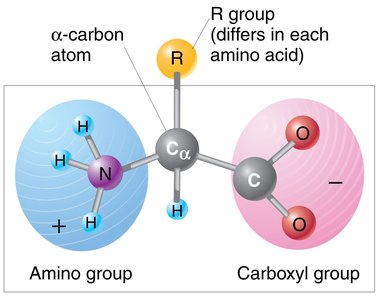
Proteins are polymers made of amino acids, each with a unique sequence. The general structure of an amino acid includes an amino group, a carboxyl group, and an R group, which varies among amino acids.
Key Point: There are 20 common amino acids found in proteins.
Key Point: The sequence and chemical properties of amino acids determine protein structure and function.
Example: Hemoglobin is a protein with a specific amino acid sequence.
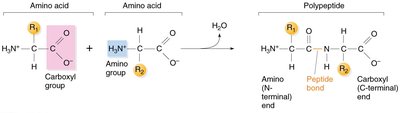
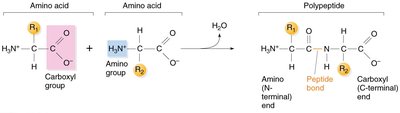
Peptide Bonds and Polypeptide Chains
Amino acids are joined by peptide bonds, forming linear chains called polypeptides. Each polypeptide has an amino terminus (start) and a carboxyl terminus (end).
Key Point: Peptide bonds are covalent bonds between the carboxyl group of one amino acid and the amino group of another.
Key Point: Polypeptide chains define the primary structure of proteins.
Levels of Protein Structure
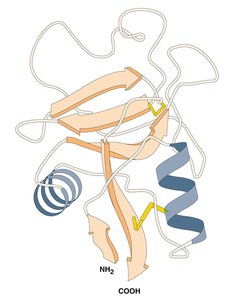
Proteins fold into precise three-dimensional structures, which determine their function. There are four levels of protein structure:
Primary: Linear sequence of amino acids.
Secondary: Local structures (alpha helix, beta sheet) formed by hydrogen bonds.
Tertiary: Final 3D shape formed by long-range interactions.
Quaternary: Association of multiple polypeptides (e.g., hemoglobin).
Enzymes: Proteins with Catalytic Function
Enzyme Structure and Function
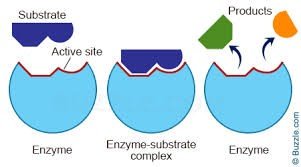
Enzymes are proteins that catalyze specific chemical reactions. They fold to create an active site, where substrates bind and reactions occur.
Key Point: Enzymes lower the activation energy of reactions.
Key Point: The active site is highly specific to the substrate.
Example: Phenylalanine hydroxylase catalyzes the breakdown of phenylalanine.
Biochemical Pathways and Genetic Diseases
Pathways and Enzyme Defects
Enzymes work together in pathways to synthesize or degrade substances. If an enzyme is missing due to a gene mutation, its substrate accumulates, leading to disease.
Key Point: Each step in a pathway is controlled by a specific enzyme encoded by a gene.
Example: Alkaptonuria and phenylketonuria (PKU) are caused by defects in enzymes of the phenylalanine breakdown pathway.
Beadle and Tatum: One Gene-One Enzyme Hypothesis
Neurospora crassa Experiments
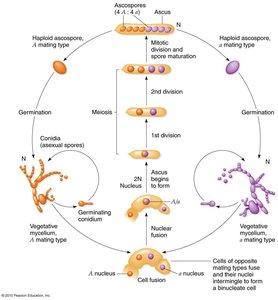
Beadle and Tatum used the bread mold Neurospora crassa to show that mutations in single genes affect specific steps in biochemical pathways. Their work led to the 'one gene-one enzyme' hypothesis, later refined to 'one gene-one polypeptide.'
Key Point: Genetic screens can identify mutants with defects in specific metabolic steps.
Example: Auxotrophic mutants require supplementation with specific amino acids or vitamins.
Human Genetic Diseases: Molecular Basis
Phenylketonuria (PKU)
PKU is caused by mutations in the gene encoding phenylalanine hydroxylase (PAH). Without PAH, phenylalanine accumulates and is converted to harmful products, leading to neurological damage.
Key Point: PKU is a monogenic disease with over 400 known mutations in PAH.
Example: Newborns are screened for PKU and placed on a low-phenylalanine diet if affected.

Tay-Sachs Disease
Tay-Sachs disease is caused by a defect in the lysosomal enzyme HEXA, leading to accumulation of its substrate and degeneration of neurons. It is common among Jewish people of European ancestry and has no cure.
Key Point: Tay-Sachs is a monogenic disease affecting neuronal function.
Example: Genetic testing has reduced prevalence in high-risk populations.
Sickle Cell Anemia: A Molecular Disease
Sickle cell anemia is caused by a mutation in the beta hemoglobin gene, resulting in a single amino acid change (E6V: glutamic acid to valine). This alters hemoglobin structure, causing red blood cells to sickle and leading to various health complications.
Key Point: Sickle cell anemia is due to a single base pair change in DNA.
Key Point: The disease is common in populations exposed to malaria due to heterozygote advantage.
Example: Protein gel electrophoresis can distinguish sickle cell genotypes.

Sickle Cell Anemia and Malaria: Population Genetics
Heterozygote Advantage
Individuals heterozygous for the sickle cell mutation (HbA/HbS) have increased resistance to malaria, leading to persistence of the mutant allele in populations where malaria is prevalent.
Key Point: Heterozygotes have higher fitness in malarial environments.
Example: The sickle cell allele is common in African, Caribbean, and Brazilian populations.
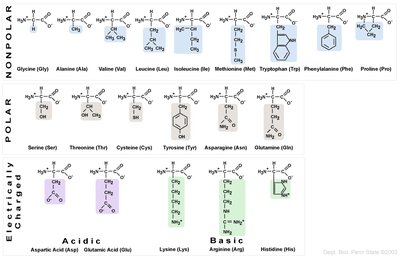
Summary Table: Amino Acid Properties
The chemical nature of the R group is used to classify amino acids into categories: nonpolar, polar, acidic, and basic.
Category | Amino Acids |
|---|---|
Nonpolar | Glycine, Alanine, Valine, Leucine, Isoleucine, Methionine, Tryptophan, Phenylalanine, Proline |
Polar | Serine, Threonine, Cysteine, Tyrosine, Asparagine, Glutamine |
Acidic | Aspartic acid, Glutamic acid |
Basic | Lysine, Arginine, Histidine |

Key Equations and Concepts
Peptide Bond Formation:
Central Dogma of Molecular Biology:
One Gene-One Polypeptide Hypothesis:
Each gene encodes a single polypeptide, which may function as an enzyme or structural protein.
