 Back
BackGenetics of Cancer: Mechanisms, Mutations, and Cellular Pathways 20
Study Guide - Smart Notes

Genetics of Cancer
Introduction to Cancer
Cancer is a group of genetic diseases characterized by uncontrolled and inappropriate cell growth. It can affect nearly all organ systems and cell types, and is the second leading cause of death in the United States. The complexity of cancer arises from its genetic basis and the diversity of tissues it affects, making it a collection of related disorders rather than a single disease.
Incidence: Over 2 million new cases and over 600,000 deaths annually in the US.
Mortality: Second only to heart disease as a cause of death.
Progress: Cancer death rates have decreased by more than 25% since the 1980s due to advances in screening, research, and treatment.

What is Cancer?
Cancer arises from the accumulation of genetic mutations that disrupt normal cellular controls. These mutations lead to the transformation of normal cells into cancerous cells, which can form tumors and, in some cases, metastasize to other parts of the body.
Benign Tumor: Remains localized and generally not life-threatening unless it compresses vital organs.
Malignant Tumor: Invades surrounding tissues and can spread (metastasize) to distant sites.
Metastatic Tumor: Secondary tumors formed by cancer cells that have migrated from the primary site.
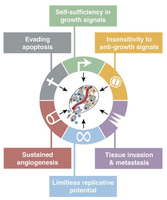
Common Properties of Cancer Cells
Despite their diversity, cancer cells share several hallmark properties that distinguish them from normal cells:
Self-sufficiency in growth signals
Insensitivity to anti-growth signals
Evading apoptosis (programmed cell death)
Sustained angiogenesis (formation of new blood vessels)
Tissue invasion and metastasis
Limitless replicative potential (immortalization)
Cellular and Molecular Mechanisms
Loss of Growth Control: Cancer cells divide without the normal regulatory signals (growth factors).
Escape from Apoptosis: Damaged cells avoid programmed cell death, allowing survival and proliferation.
Transformed Phenotype: Cancer cells exhibit altered morphology, loss of contact inhibition, anchorage independence, and immortality in culture.
Angiogenesis: Cancer cells stimulate the growth of new blood vessels to supply nutrients to the tumor.
Metastasis: Cancer cells invade surrounding tissues and spread to distant organs via the bloodstream or lymphatic system.
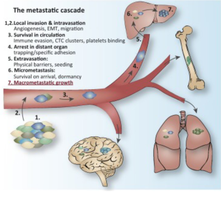
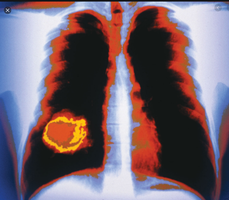
Genetic Basis of Cancer
Cancer is primarily a genetic disease caused by mutations in the genome. These mutations can be somatic (acquired) or germline (inherited):
Sporadic Cancer: ~90% of cases, mutations arise in somatic cells during a person's lifetime.
Hereditary Cancer: ~10% of cases, mutations are present in all cells due to inheritance from a parent.
The multi-hit model of carcinogenesis states that multiple mutations in key genes are required for a cell to become cancerous. Tumors are typically clonal, arising from a single progenitor cell that has accumulated these mutations.
Environmental and Lifestyle Factors
Both genetic predisposition and environmental exposures contribute to cancer risk. Most carcinogens are mutagens that increase the frequency of mutations in the genome.
Chemicals: Tobacco, industrial chemicals, and dietary factors (e.g., red meat) are linked to specific cancers.
Radiation: UV light is a major cause of skin cancer.
Infectious Agents: Certain viruses (e.g., HPV) and bacteria (e.g., Helicobacter pylori) are associated with cancer development.
Hereditary Cancer Syndromes
Germline mutations in specific genes can greatly increase cancer risk. Examples include:
BRCA1/BRCA2: Mutations predispose to breast and ovarian cancer.
Retinoblastoma (RB1 gene): Inherited mutations lead to a high risk of bilateral eye tumors in children.
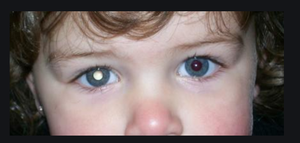
Knudson's two-hit hypothesis explains that both alleles of a tumor suppressor gene must be inactivated for cancer to develop. In hereditary cases, one allele is already mutated in all cells, so only one additional mutation is needed.
Types of Genes Mutated in Cancer
Oncogenes: Gain-of-function mutations in proto-oncogenes (e.g., Ras) lead to uncontrolled cell division. Only one mutated copy is needed (dominant effect).
Tumor Suppressor Genes: Loss-of-function mutations (e.g., p53, RB1) remove critical cell cycle checkpoints. Both copies must be mutated (recessive effect).
DNA Repair Genes: Mutations in these genes (e.g., BRCA1/2, XP genes) increase the overall mutation rate, predisposing to cancer.
Telomerase: Reactivation in cancer cells allows limitless replication by maintaining telomere length.
Key Molecular Pathways in Cancer
Growth Factor Signaling: Mutations in genes like Ras can lock signaling pathways in the "on" state, driving cell division without external signals.
Cell Cycle Checkpoints: Tumor suppressors like p53 monitor DNA integrity and can halt the cell cycle or trigger apoptosis if damage is detected.
DNA Damage Response: Defects in DNA repair genes lead to genomic instability and accumulation of mutations.
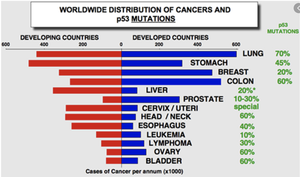
Immortalization and Telomerase
Normal somatic cells have a limited capacity to divide (the Hayflick limit) due to progressive telomere shortening. Most cancer cells reactivate telomerase, enabling them to divide indefinitely and become "immortalized." This is a key feature of cancer cell lines used in research.
Summary Table: Types of Genes Mutated in Cancer
Gene Type | Normal Function | Effect of Mutation | Inheritance Pattern |
|---|---|---|---|
Proto-oncogene | Promote cell division in response to signals | Gain-of-function (oncogene): uncontrolled division | Dominant (one allele) |
Tumor suppressor | Inhibit cell division, promote apoptosis, repair DNA | Loss-of-function: loss of growth control | Recessive (both alleles) |
DNA repair gene | Repair DNA damage | Loss-of-function: increased mutation rate | Recessive (both alleles) |
Telomerase | Maintain telomere length | Reactivation: limitless replication | Usually somatic activation |
Conclusion
Cancer is fundamentally a genetic disease resulting from the accumulation of mutations in key regulatory genes. Both inherited and environmental factors contribute to cancer risk. Understanding the molecular genetics of cancer is essential for developing targeted therapies and preventive strategies.
