 Back
BackMembrane Transport Proteins and Ion Channels: Structure, Function, and Genetic Relevance
Study Guide - Smart Notes

Membrane Transport Proteins
Overview of Membrane Transport
Cell membranes are selectively permeable barriers that regulate the movement of substances into and out of cells. Most molecules cannot diffuse freely across the plasma membrane due to the hydrophobic nature of the lipid bilayer. Specialized membrane transport proteins facilitate the passage of select substances, ensuring proper cellular function and homeostasis.
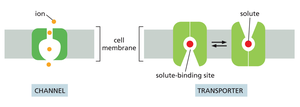
Transporters: Move small organic molecules or inorganic ions across the membrane by binding them specifically and undergoing conformational changes.
Channels: Form hydrophilic pores that allow specific ions or water molecules to diffuse across the membrane.
Ion Channels: Permit passage of inorganic ions only, contributing to the generation and regulation of membrane potential.
Lipid Bilayer Permeability
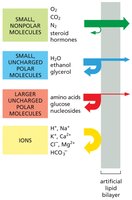
The lipid bilayer is inherently impermeable to ions and most uncharged polar molecules. The rate at which a molecule diffuses across a protein-free artificial lipid bilayer depends on its size and solubility:
Smaller and more hydrophobic (nonpolar) molecules diffuse more rapidly.
Polar, water-soluble solutes require transport proteins to cross the bilayer.
Charged molecules are highly impermeable without assistance.
Ion Concentrations and Membrane Potential
Ion Gradients Across Membranes
Cells maintain distinct ion concentrations inside and outside the plasma membrane, which is essential for many physiological processes. The impermeability of the membrane to inorganic ions allows cells to establish and maintain these gradients.
Extracellular fluid: High Na+, balanced by Cl-
Intracellular fluid: High K+, balanced by negatively charged ions
Cation/Anion | Intracellular Concentration (mM) | Extracellular Concentration (mM) |
|---|---|---|
Na+ | 5–15 | 145 |
K+ | 140 | 5 |
Mg2+ | 0.5 | 1–2 |
Ca2+ | 10–4 | 1–2 |
H+ | 7 × 10–5 (pH 7.2) | 4 × 10–5 (pH 7.4) |
Cl– | 5–15 | 110 |

Membrane Potential
The membrane potential is the voltage difference across the plasma membrane, resulting from small excesses of positive or negative charge concentrated near the membrane. In unstimulated cells, the exchange of anions and cations is balanced, resulting in a resting membrane potential (typically –20 to –200 mV in animal cells). This potential powers the transport of some metabolites and is crucial for electrical signaling in cells.
Types of Membrane Transport Proteins
Channels vs. Transporters
Membrane transport proteins are highly selective, allowing passage of only specific molecules or ions. There are two main classes:
Channels: Discriminate mainly by size and charge; open channels allow any appropriately sized and charged ion or molecule to pass through.
Transporters: Bind solutes with high specificity and transfer only those that are specifically bound, similar to enzyme-substrate interactions.
Passive and Active Transport
Solutes cross membranes by either passive or active transport:
Passive transport: Spontaneous movement down a concentration gradient (all channels and some transporters).
Active transport: Movement against a concentration gradient, requiring energy input (only transporters, known as pumps).
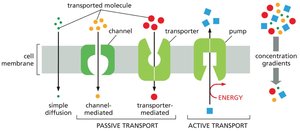
Passive Transport and Osmosis
Electrochemical Gradient
The electrochemical gradient is the net force driving a charged molecule across a membrane, determined by both the concentration gradient and the membrane potential. This gradient dictates the direction of passive transport for each solute.
Water Transport and Osmosis
Water moves passively across cell membranes, but the process is slow. Specialized channels called aquaporins facilitate rapid and selective water movement. The osmolarity of solutes inside the cell typically exceeds that outside, creating an osmotic gradient that pulls water into the cell. The movement of water down its concentration gradient is known as osmosis.
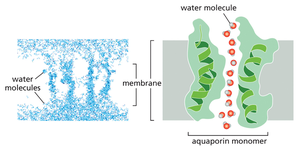
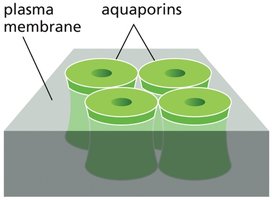
Transporters and Pumps
Specificity and Distribution of Transporters
Transporters are responsible for the movement of most small, water-soluble organic and some inorganic molecules across cell membranes. Each transporter is highly selective, and each cell membrane contains a characteristic set of transporters tailored to its function.
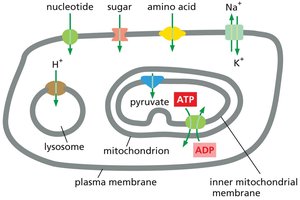
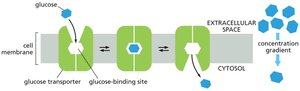
Passive Transporters
Passive transporters, such as the glucose transporter, move solutes along their electrochemical gradients without determining the direction of transport. The direction is dictated solely by the concentration gradient.
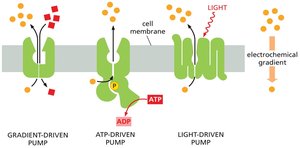
Active Transport and Pumps
Active transport is essential for maintaining correct intracellular solute concentrations. There are three main types of pumps:
Gradient-driven pumps: Couple the uphill transport of one solute to the downhill transport of another.
ATP-driven pumps: Use energy from ATP hydrolysis to drive uphill transport.
Light- or redox-driven pumps: Use energy from sunlight or redox reactions (found in bacteria, archaea, and mitochondria).
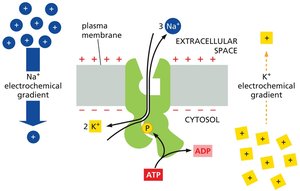
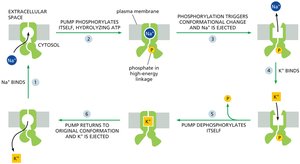
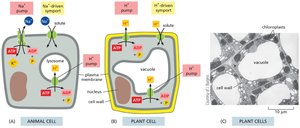
Na+-K+ ATPase (Na+ Pump)
The Na+ pump uses ATP hydrolysis to pump Na+ out of animal cells and K+ in, maintaining low cytosolic Na+ and high K+. This pump accounts for about 30% of total ATP consumption in animal cells and is crucial for maintaining membrane potential and secondary active transport.
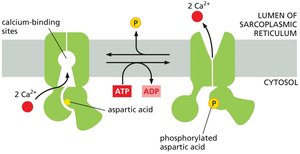
Ca2+ Pump
Ca2+ pumps keep cytosolic Ca2+ concentrations low, which is essential for cellular signaling, such as muscle contraction. These pumps are ATPases similar to the Na+ pump but do not require transporting a second ion.
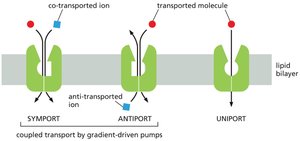
Gradient-Driven Pumps: Symport, Antiport, Uniport
Gradient-driven pumps use the movement of one solute down its gradient to power the uphill transport of another. Types include:
Symport: Both solutes move in the same direction.
Antiport: Solutes move in opposite directions.
Uniport: Only one solute is transported (not coupled).
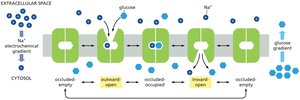
Glucose-Na+ Symport
The electrochemical Na+ gradient drives the transport of glucose across the plasma membrane of animal cells. The glucose-Na+ symport enables cells to take up glucose from the gut even when its concentration is higher inside the cell than in the gut lumen.
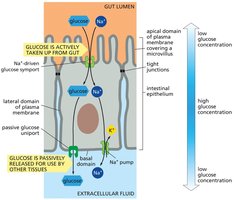
Glucose Transport in Epithelial Cells
Two types of glucose transporters enable gut epithelial cells to transfer glucose across the epithelial lining: a symport at the apical domain for uptake and a passive uniport at the basal and lateral domains for release. Tight junctions keep these transporters separated.
H+ Gradients in Plants, Fungi, and Bacteria
Plant, bacterial, and fungal cells rely on electrochemical gradients of H+ (protons) for solute import, as they lack Na+ pumps in their plasma membranes. ATP-dependent H+ pumps in organelle membranes maintain acidic environments within organelles.
Ion Channels: Structure and Function
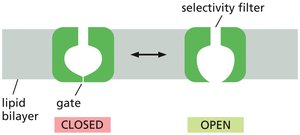
Ion Selectivity and Gating
Ion channels are selective for specific ions, determined by the diameter, shape, and charge distribution of the channel. They are not continuously open but are gated, opening and closing in response to specific stimuli. Ion channels allow rapid ion flow down electrochemical gradients but cannot perform active transport.
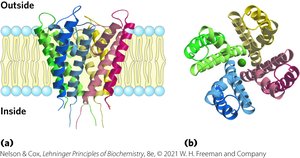
K+ Channel Specificity
The structure of K+ ion channels reveals that the channel is just wide enough to accommodate a dehydrated K+ ion, allowing it to pass 10,000 times more readily than Na+. The selectivity filter precisely fits K+, and smaller ions cannot compensate for the energy of desolvation.
Membrane Potential and Ion Channel Regulation
Generation of Membrane Potential
Membrane potential is governed by the permeability of the membrane to specific ions. When ions of one type cross the membrane, they set up a charge difference, creating a membrane potential. Only a small fraction of ions must move to establish this potential.
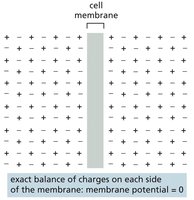
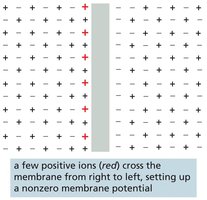
K+ Leak Channels and Resting Membrane Potential
K+ leak channels play a major role in generating the resting membrane potential in animal cells. When open, K+ flows out of the cell, leaving behind unbalanced negative charges and generating a voltage difference. The resulting imbalance opposes further K+ movement, stabilizing the membrane potential.
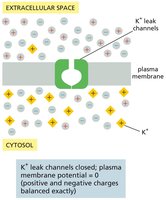
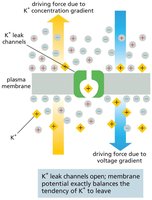
Patch-Clamp Recording and Ion Channel Activity
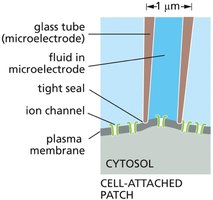
Patch-Clamp Technique
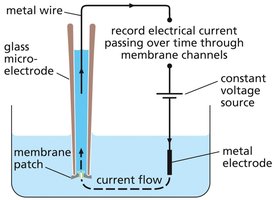
Patch-clamp recording is a powerful technique used to monitor the activity of individual ion channels. A glass microelectrode forms a tight seal with the cell membrane, allowing measurement of electrical currents through single channels and manipulation of ion concentrations and membrane potential.

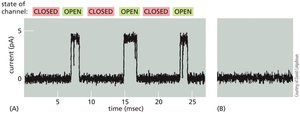
Ion Channel Gating and Regulation
Ion channels randomly transition between open and closed states in an all-or-none manner. Their activity can be regulated by various stimuli, which bias the probability of the channel being open or closed.
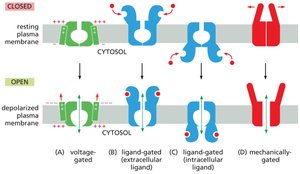
Types of Ion Channel Gating
There are over 200 types of ion channels, differing in ion specificity and gating mechanisms:
Voltage-gated: Open in response to changes in membrane potential.
Ligand-gated: Open in response to binding of a ligand (extracellular or intracellular).
Mechanically-gated: Open in response to mechanical force.
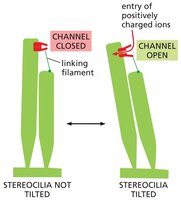
Mechanically-Gated Ion Channels in Hearing
Mechanically-gated ion channels are essential for hearing. In auditory hair cells, sound vibrations pull the channels open, allowing ions to flow in and generate electrical signals transmitted to the auditory nerve.
Voltage-Gated Ion Channels and Electrical Signaling
Voltage-gated ion channels respond to changes in membrane potential, with voltage sensor domains that trigger opening above a threshold. The opening of one type of voltage-gated channel can alter the membrane potential, activating or inactivating other channels. This control circuit is fundamental to all electrical signaling in cells.
Ion Channels in Plant Responses
Mechanically-gated and voltage-gated ion channels underlie rapid plant responses, such as the leaf-closing reaction in Mimosa pudica. Mechanical stimulation opens channels, generating an electrical impulse that leads to water loss in hinge cells and leaf folding.

Additional info: Understanding membrane transport proteins and ion channels is fundamental to genetics, as these processes underlie cellular homeostasis, signaling, and the genetic control of physiological traits. Mutations in genes encoding these proteins can lead to a variety of genetic disorders, emphasizing their importance in the field of genetics.
