 Back
BackMutation, DNA Repair, and Homologous Recombination
Study Guide - Smart Notes

Mutation: Overview and Types
Definition and Importance
Mutations are heritable, permanent changes in the DNA sequence of an organism's genome. They are the original source of genetic diversity, fueling evolutionary change, but are usually rare and often deleterious. Mutation rates vary widely among organisms and even among genes within a species, influenced by genome size and life cycle.
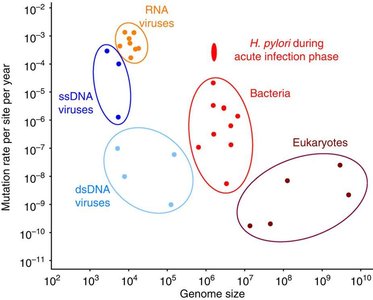
Mutation Rate: Ranges from as low as to as high as mutations per nucleotide per year, depending on the organism.
RNA viruses have high mutation rates due to lack of proofreading by RNA-dependent RNA polymerases.
Bacteria and eukaryotes generally have lower mutation rates, with exceptions such as H. pylori during acute infection.
Types of Mutations
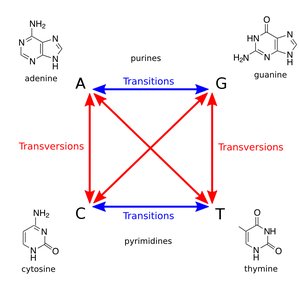
Point Mutations: Change in a single nucleotide or a few nucleotides.
Transition: Purine replaces purine (A ↔ G) or pyrimidine replaces pyrimidine (C ↔ T).
Transversion: Purine replaces pyrimidine or vice versa (A/G ↔ C/T).
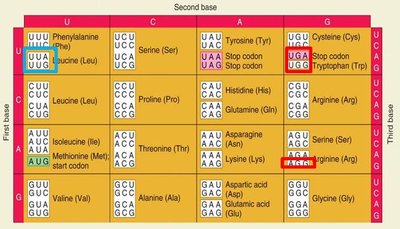
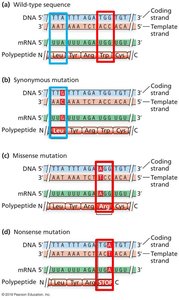
Point Mutations in Coding Regions
Silent (synonymous): No change in amino acid.
Missense: Change in amino acid.
Nonsense: Change to a stop codon.
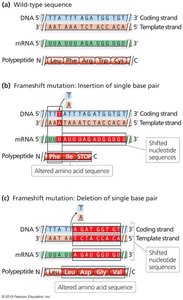
Frameshift Mutations
Frameshift mutations result from insertions or deletions (indels) that change the reading frame, often introducing premature stop codons. Not all indels cause frameshifts; only those not in multiples of three nucleotides do.
Regulatory Mutations
Some point mutations affect gene expression levels rather than protein sequence. These can occur in promoters, introns, or untranslated regions (UTRs), affecting splicing, polyadenylation, or cryptic splice site creation.
Forward Mutation and Reversion
Forward mutation: Wild-type allele → mutant allele.
Reverse mutation (reversion): Mutant allele → wild-type or near wild-type allele.
Suppressor mutation: A second mutation suppresses the effect of the first, potentially restoring function.
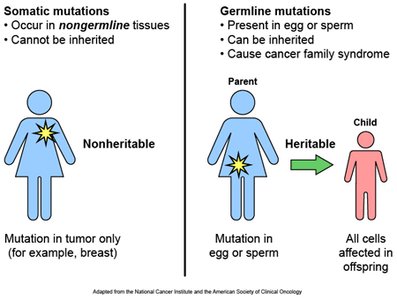
Somatic vs. Germ Line Mutations
Somatic mutations: Occur in non-reproductive tissues; not inherited.
Germ-line mutations: Occur in reproductive cells; can be passed to offspring.
Mechanisms of Mutation: Spontaneous vs. Induced
Spontaneous Mutations
Spontaneous mutations arise without external influence, mainly due to errors in DNA replication or spontaneous chemical changes in DNA bases.
Repeat mutations: Caused by strand slippage during DNA replication, leading to expansion or contraction of repeat sequences.
Tautomeric shifts: Temporary changes in base structure lead to mispairing during replication.
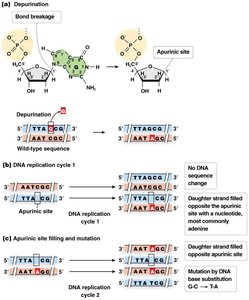
Depurination: Loss of a purine base, creating an apurinic site.
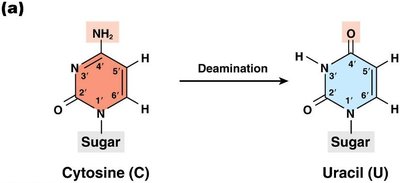
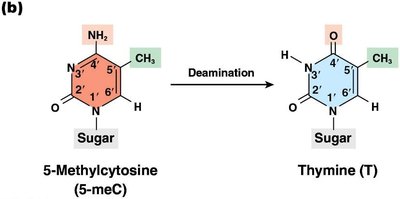
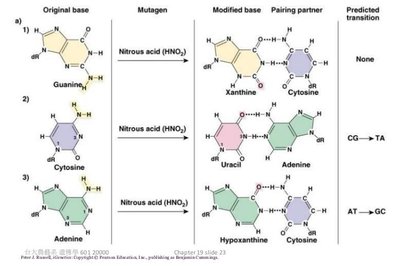
Deamination: Removal of an amino group, often converting cytosine to uracil.
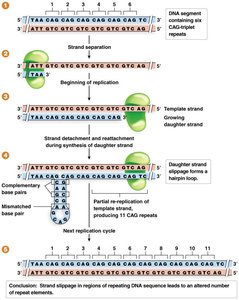
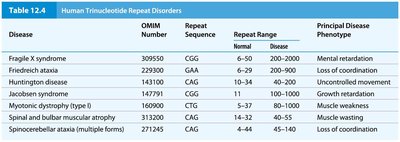
Trinucleotide Repeat Disorders
Expansion of trinucleotide repeats beyond a threshold causes several human genetic disorders.
Disease | Repeat Sequence | Repeat Range (Normal) | Repeat Range (Disease) | Principal Phenotype |
|---|---|---|---|---|
Fragile X syndrome | CGG | 6-50 | 200-2000 | Mental retardation |
Friedreich ataxia | GAA | 7-34 | 200-900 | Loss of coordination |
Huntington disease | CAG | 10-34 | 40-100 | Uncontrolled movement |
Jacobsen syndrome | CGG | 14-60 | 100-1000 | Growth retardation |
Myotonic dystrophy (type 1) | CTG | 5-37 | 80-1000 | Muscle weakness |
Spinal and bulbar muscular atrophy | CAG | 11-32 | 38-62 | Muscle wasting |
Spinocerebellar ataxia | CAG | 4-44 | 45-140 | Loss of coordination |
Tautomeric Shifts
Purines and pyrimidines can exist in rare tautomeric forms, leading to abnormal base pairing during replication and resulting in point mutations.
Depurination
Depurination is the loss of a purine base (adenine or guanine) from DNA, creating an apurinic site. DNA polymerase often inserts an adenine opposite the site during replication, potentially causing mutations.
Deamination
Deamination is the removal of an amino group from a nucleotide. For example, deamination of cytosine produces uracil, which pairs with adenine, resulting in a C to T transition mutation. Deamination of 5-methylcytosine produces thymine, creating a G:T mispair.
Induced Mutations: Chemical and Physical Mutagens
Chemical Mutagens
Deaminating agents: Remove amine groups (e.g., nitrous acid).
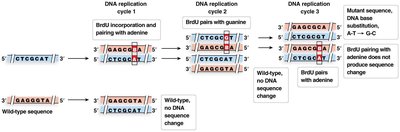
Nucleotide base analogs: Mimic normal bases but pair incorrectly (e.g., BrdU).
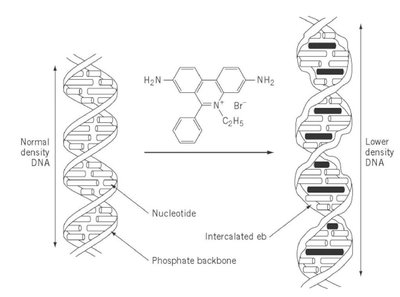
Intercalating agents: Insert between base pairs, causing frameshifts (e.g., ethidium bromide).
Oxidizing, alkylating, and hydroxylating agents: Modify bases, leading to mispairing or strand breaks.
Physical Mutagens
UV light: Causes pyrimidine dimers and photoproducts, distorting DNA and blocking replication.
X-rays and radioactive materials: Cause single- and double-strand breaks, base loss, and cross-linking.
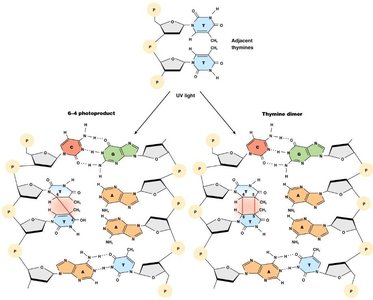
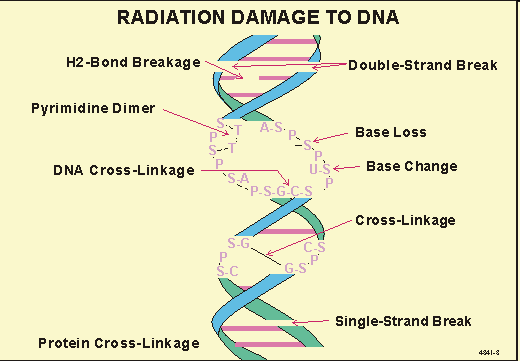
DNA Repair Mechanisms
Overview
Both prokaryotes and eukaryotes have evolved multiple DNA repair pathways to maintain genome stability. These include base excision repair (BER), mismatch repair (MMR), nucleotide excision repair (NER), direct reversal, and double-strand break repair (DSB).
Base Excision Repair (BER)
Removes damaged or incorrect bases (e.g., deaminated cytosine).
Involves DNA glycosylases, AP endonuclease, DNA polymerase, and DNA ligase.
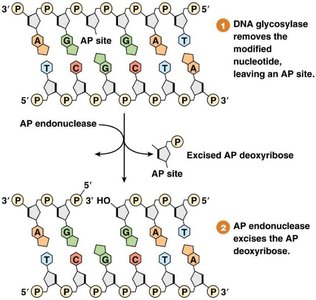
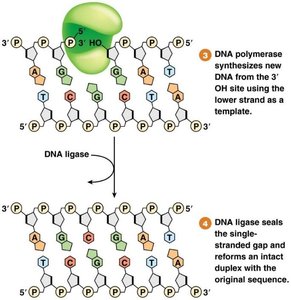
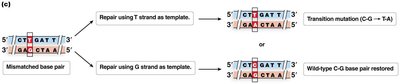
Mismatch Repair (MMR)
Corrects mismatched base pairs after replication.
Prokaryotes use methylation to distinguish old and new strands; eukaryotes use nicks in the new strand.
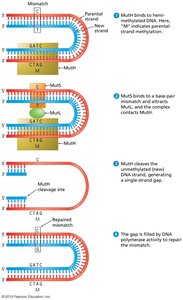
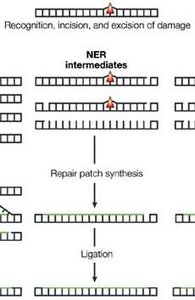
Nucleotide Excision Repair (NER)
Removes bulky DNA lesions, such as those caused by UV light.
Enzymes excise a short stretch of nucleotides containing the lesion; DNA polymerase and ligase fill and seal the gap.
Direct Reversal
Photoreactivation directly reverses UV-induced pyrimidine dimers using photolyase (not present in placental mammals).
Double-Strand Break Repair (DSB)
Nonhomologous End Joining (NHEJ): Directly ligates broken DNA ends, often with loss of nucleotides.
Synthesis-Dependent Strand Annealing (SDSA): Uses homologous recombination for error-free repair, especially in dividing cells.
Homologous Recombination
Mechanism and Biological Role
Homologous recombination is the exchange of genetic material between homologous DNA molecules, crucial for double-strand break repair and meiotic crossing over. In eukaryotes, it occurs during prophase I of meiosis and is essential for genetic diversity.
Initiated by double-strand breaks (e.g., by SPO11 in meiosis).
Strand invasion and D-loop formation mediated by proteins such as Rad51.
Resolution of Holliday junctions determines crossover or non-crossover products.
Gene conversion can result from mismatch repair in heteroduplex DNA.
The Ames Test: Detecting Mutagenicity
Principle and Application
The Ames test is a rapid bacterial assay to determine if a chemical is mutagenic. It uses Salmonella typhimurium strains that cannot synthesize histidine due to specific mutations. Exposure to a mutagen may cause reversion to wild-type, allowing growth on histidine-free media.
Tested compounds may require metabolic activation (e.g., by S9 liver extract) to become mutagenic.
Revertant colonies indicate mutagenicity; the test is widely used for screening potential carcinogens.
Summary: Key Points
Mutations are rare, random, and the source of genetic diversity.
They can be spontaneous or induced by chemical/physical agents.
Cells have evolved multiple repair mechanisms to maintain genome integrity.
Homologous recombination is essential for repair and genetic diversity.
The Ames test is a practical tool for detecting mutagenic compounds.
