 Back
BackProteomics, Genomics, Bioinformatics, and Cancer Genetics: Advanced Topics in Modern Genetics
Study Guide - Smart Notes

Proteomics: Protein Structure and Function
Levels of Protein Structure
Proteins are complex macromolecules essential for cellular structure and function. Their structure is organized into four hierarchical levels:
Primary Structure: The linear sequence of amino acids in a polypeptide chain, determined by the gene encoding the protein.
Secondary Structure: Local folding patterns stabilized by hydrogen bonds, including the alpha helix and beta-pleated sheet.
Tertiary Structure: The overall three-dimensional conformation of a single polypeptide chain, resulting from interactions among R-groups.
Quaternary Structure: The association of multiple polypeptide chains (subunits) into a functional protein complex.
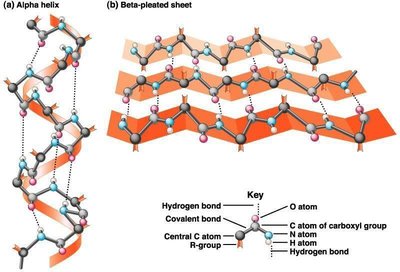
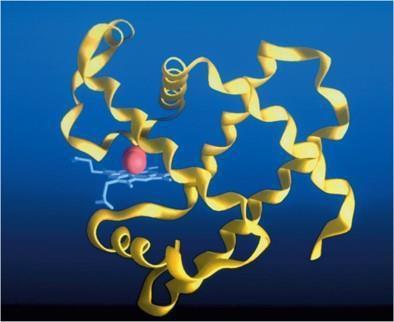
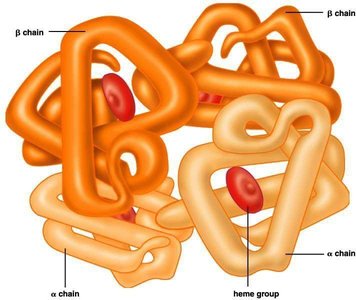
Diversity of Protein Functions
Immunoglobulins: Antibodies that function in the immune response of vertebrates.
Transport Proteins: Facilitate movement of molecules across cellular membranes (e.g., hemoglobin).
Hormones and Receptors: Regulate physiological processes by signaling and response mechanisms.
Histones: Bind to DNA and play a role in chromatin structure in eukaryotes.
Transcription Factors: Regulate gene expression by binding to specific DNA sequences.
Enzymes
Enzymes are proteins that catalyze biochemical reactions, increasing reaction rates and determining the metabolic capacity of cells. They are highly specific and essential for life.
Sickle-Cell Anemia: A Molecular Disease
Sickle-cell anemia is a recessive genetic disorder caused by a mutation in the beta-globin gene of hemoglobin (HbS allele). Homozygous individuals exhibit sickle-shaped erythrocytes under low oxygen tension, leading to various health complications. Heterozygotes are typically asymptomatic carriers.
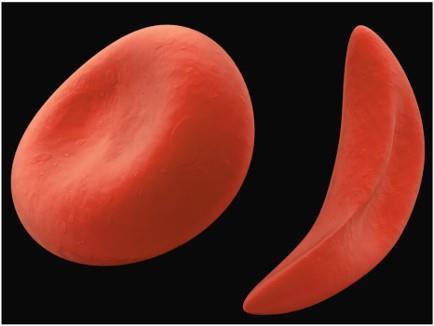

Example: The substitution of valine for glutamic acid at position 6 in the beta chain of hemoglobin causes the sickling phenotype.
Genomics and Bioinformatics
Polymerase Chain Reaction (PCR)
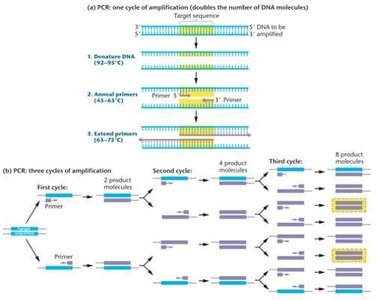
PCR is a rapid, in vitro method for amplifying specific DNA sequences, eliminating the need for host cells in cloning. It is widely used in research, diagnostics, and forensics.
Requirements: Double-stranded target DNA, DNA polymerase, Mg2+ ions, four deoxyribonucleoside triphosphates, and two primers (complementary to the 5' and 3' ends of the target sequence).
Applications: Amplification from minimal DNA sources such as dried blood, semen, or hair.
Genomics
Genomics is the comprehensive study of genomes, providing insights into gene structure, function, and evolution. It is one of the fastest-growing fields in genetics.
Bioinformatics
Bioinformatics utilizes mathematical and computational tools to organize, analyze, and interpret biological data, including gene sequences, expression profiles, and protein structures.
Applications include comparing DNA sequences, identifying genes and regulatory regions, predicting protein sequences, and deducing evolutionary relationships.
Next-Generation Sequencing (NGS)
NGS technologies enable simultaneous sequencing of thousands of DNA molecules, generating vast amounts of data quickly and cost-effectively using fluorescence imaging.
GenBank and Genome Databases
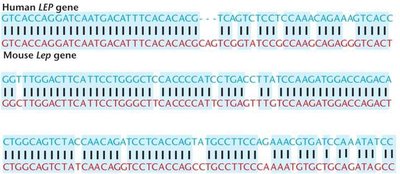
GenBank, maintained by the NCBI, is the largest public DNA sequence database. Each entry receives a unique accession number for retrieval and analysis. Comparative genomics can reveal evolutionary conservation between species, as seen in the similarity between human LEP and mouse Lep genes.
The Genetics of Cancer
Cancer as a Genetic Disease
Cancer arises from genetic alterations in somatic cells, including point mutations, chromosomal rearrangements, amplifications, and deletions. Only a small percentage of cancers are due to inherited (germ-line) mutations.
Chromosomal Translocations in Cancer
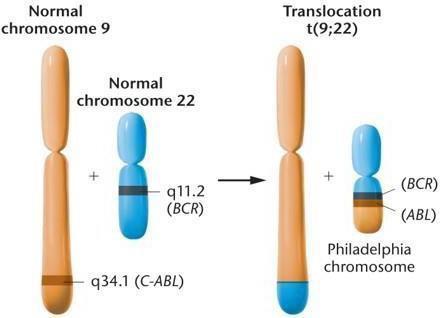
Reciprocal chromosomal translocations are common in cancers such as leukemias and lymphomas. For example, the Philadelphia chromosome in chronic myelogenous leukemia (CML) results from a t(9;22) translocation, creating a fusion gene that drives uncontrolled cell division.
Properties of Cancer Cells
Proliferation: Uncontrolled cell growth and division.
Metastasis: Spread of cancer cells to other tissues, forming secondary tumors.
Benign Tumors: Non-invasive, localized growths.
Malignant Tumors: Invasive, metastatic, and life-threatening.
The cancer stem cell hypothesis suggests that only a subset of tumor cells (cancer stem cells) can initiate new tumors due to their self-renewal capacity.
Multistep Nature of Cancer Development
Cancer develops through the accumulation of multiple mutations over time, often taking decades to progress from normal cells to malignant tumors. This is exemplified in colorectal cancer, where sequential driver mutations lead to tumorigenesis.
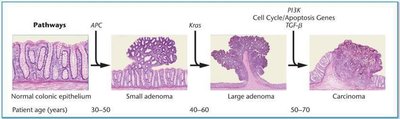
Genomic Instability and Mutator Phenotype
Cancer cells exhibit high rates of mutation and chromosomal abnormalities, including translocations, aneuploidy, and deletions. This genomic instability increases the likelihood of acquiring additional cancer-promoting mutations.
Proto-oncogenes and Tumor-Suppressor Genes
Proto-oncogenes promote cell growth and division; mutations can convert them into oncogenes, driving cancer. Tumor-suppressor genes inhibit cell division or promote apoptosis; their inactivation removes growth restraints.
Gene | Normal Function | Alteration in Cancer | Associated Cancers |
|---|---|---|---|
c-myc | Transcription factor, regulates cell cycle, differentiation, apoptosis | Translocation, amplification, point mutations | Lymphomas, leukemias, lung cancer, many types |
c-kit | Tyrosine kinase, signal transduction | Mutation | Sarcomas |
RARα | Hormone-dependent transcription factor, differentiation | Chromosomal translocations with PML gene, fusion product | Acute promyelocytic leukemia |
Cyclins | Bind to CDKs, regulate cell cycle | Gene amplification, overexpression | Lung, esophagus, many types |
RB1 | Cell-cycle checkpoints, binds E2F | Mutation, deletion, inactivation by viral oncogene products | Retinoblastoma, osteosarcoma, many types |
TP53 | Transcription regulation | Mutation, deletion, viruses | Many types |
BRCA1, BRCA2 | DNA repair | Point mutations | Breast, ovarian, prostate cancers |
TP53: The Guardian of the Genome
TP53 encodes the p53 protein, a transcription factor that regulates cell cycle arrest and apoptosis in response to DNA damage. Mutations in TP53 are found in about 50% of all cancers, leading to loss of cell cycle control and increased mutation rates.
Inherited Cancer Predisposition Syndromes
Tumor Predisposition Syndrome | Chromosome | Gene Affected |
|---|---|---|
Early-onset familial breast cancer | 17q | BRCA1 |
Familial adenomatous polyposis | 5q | APC |
Familial melanoma | 9p | CDKN2 |
Gorlin syndrome | 9q | PTCH1 |
Hereditary nonpolyposis colon cancer | 2p | MSH2, 6 |
Li-Fraumeni syndrome | 17p | TP53 |
Multiple endocrine neoplasia, type 1 | 11q | MEN1 |
Multiple endocrine neoplasia, type 2 | 10q | RET |
Neurofibromatosis, type 1 | 17q | NF1 |
Neurofibromatosis, type 2 | 22q | NF2 |
Retinoblastoma | 13q | pRb |
Von Hippel–Lindau syndrome | 3p | VHL |
Wilms tumor | 11p | WT1 |
Carcinogens and Cancer Risk
Carcinogens are agents that cause cancer, often with a long latency period. Examples include aflatoxin, nitrosamines, tobacco smoke, and certain viruses. Tobacco smoke is associated with multiple cancer types and induces both genetic and epigenetic changes.
Viruses and Cancer
Virus | Associated Cancers |
|---|---|
Epstein-Barr virus (EBV) | Burkitt lymphoma, nasopharyngeal carcinoma, Hodgkin lymphoma |
Hepatitis B virus (HBV) | Hepatocellular carcinoma |
Hepatitis C virus (HCV) | Hepatocellular carcinoma, non-Hodgkin lymphoma |
Human papilloma viruses 16, 18 (HPV16, 18) | Cervical cancer, anogenital cancers, oral cancers |
Kaposi sarcoma–associated herpesvirus (KSHV) | Kaposi sarcoma, primary effusion lymphoma |
Human T-cell lymphotropic virus, type 1 (HTLV-1) | Adult T-cell leukemia and lymphoma |
Human immunodeficiency virus, type 1 (HIV-1) | Immune suppression, leading to cancers caused by other viruses (KSHV, EBV, HPV) |
Genome Editing with CRISPR-Cas
CRISPR-Cas9 Technology
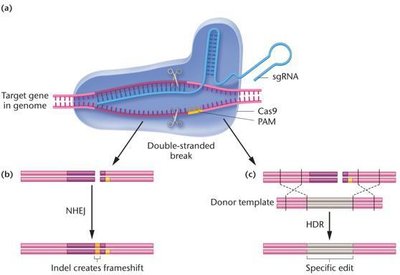
Genome editing involves the precise removal, addition, or alteration of DNA sequences in living cells. The CRISPR-Cas9 system, adapted from bacterial immune defense, is the most efficient and versatile genome editing tool.
An sgRNA guides the Cas9 nuclease to a specific DNA sequence adjacent to a PAM site, where it introduces a double-stranded break.
Repair can occur via non-homologous end joining (NHEJ), introducing indels, or homology-directed repair (HDR), allowing precise edits using a donor template.
Applications of CRISPR-Cas
Crop improvement (e.g., faster-ripening tomatoes, enhanced nutritional traits)
Gene therapy for genetic diseases and cancer (clinical trials underway)
Potential de-extinction projects (e.g., woolly mammoth)
Livestock disease resistance
