 Back
BackRecombinant DNA Technology and Transgenic Models: Applications and Methods
Study Guide - Smart Notes

Recombinant DNA Technology: Applications and Methods
Overview of Recombinant DNA Technology
Recombinant DNA technology involves the experimental manipulation of DNA in the laboratory, including the addition, deletion, or modification of genes. This technology has revolutionized genetics by enabling the study and application of gene function in various organisms, including the creation of transgenic models.
Industrial Applications: Production of enzymes and hormones for therapeutic use.
Clinical Applications: Identification of disease-causing mutations, gene therapy, and cataloging genetic polymorphisms.
Basic Research: Analysis of gene expression, protein function, and regulatory mechanisms.
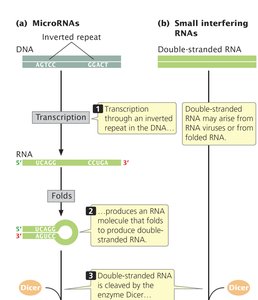
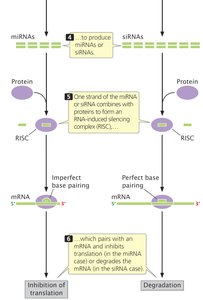
Small Interfering RNAs (siRNAs) and MicroRNAs (miRNAs)
Small interfering RNAs and microRNAs are short RNA molecules that regulate gene expression post-transcriptionally. They are produced when the enzyme Dicer cleaves larger double-stranded RNA molecules.
Functions: mRNA degradation, inhibition of translation, DNA methylation, and chromatin remodeling.
siRNAs: Artificially synthesized, 19–23 nucleotides, used for transient gene silencing.
miRNAs: Endogenously produced, regulate gene expression by imperfect base pairing with target mRNAs.
Plasmids and Vectors in Recombinant DNA Technology
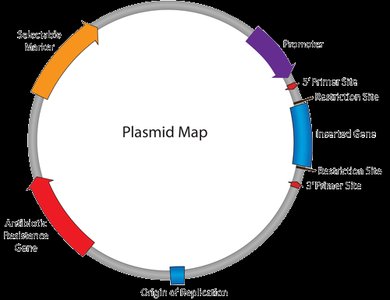
Structure and Function of Plasmids
Plasmids are double-stranded, circular DNA molecules capable of independent replication in bacteria. They are essential tools for gene cloning and expression.
Key Elements: Origin of replication (ori), antibiotic resistance gene, multiple cloning sites, and promoter.
Promoters: Constitutive (always active) or inducible (activated by stimuli).
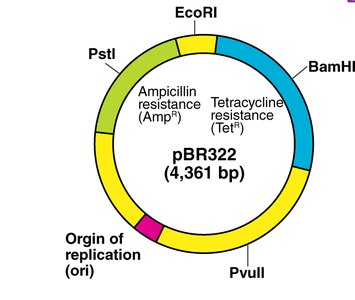
Example: Plasmid pBR322
pBR322 is a commonly used plasmid vector containing two antibiotic resistance genes (ampicillin and tetracycline) and multiple restriction sites for cloning.
Selection: Only bacteria with the plasmid survive antibiotic treatment.
Insertional Inactivation: Insertion of foreign DNA into an antibiotic resistance gene disrupts its function, allowing for identification of recombinant clones.
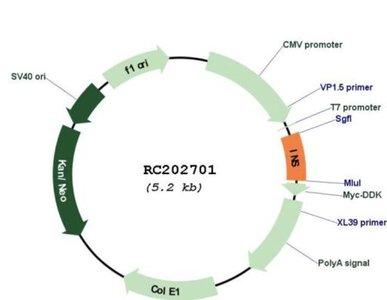
Gene Overexpression Vectors
Vectors designed for gene overexpression in eukaryotic cells often include strong promoters (e.g., CMV promoter) and selectable markers.
Gene Transfer Methods
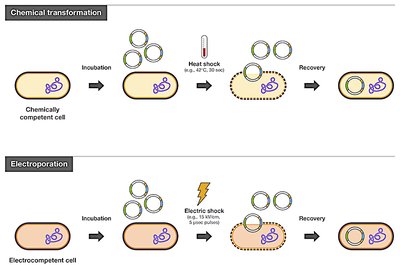
Bacterial Transformation
Transformation is the process of introducing foreign DNA into bacterial cells. Common methods include chemical transformation (heat shock) and electroporation.
Competent Cells: Bacteria made permeable to DNA uptake (e.g., DH5α, XL10-Gold).
Selection: Transformed cells are selected using antibiotic-containing media.
Gene Transfer in Eukaryotes: Transfection
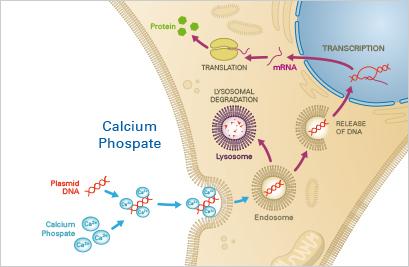
Transfection refers to the introduction of foreign DNA into eukaryotic cells using chemical or physical methods.
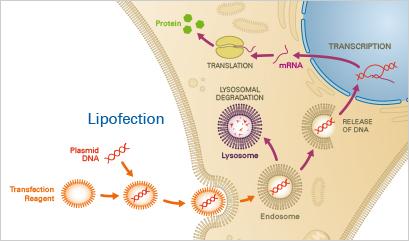
Chemical Methods: Calcium phosphate co-precipitation, cationic polymers, and lipofection.
Lipofection: DNA-lipid complexes facilitate DNA entry via endocytosis.
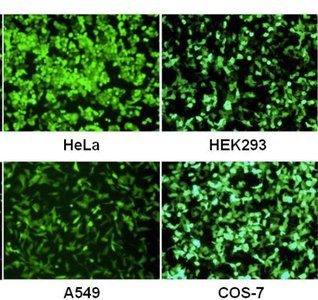
Gene Transfer in Eukaryotes: Viral Vectors
Viral vectors, such as lentiviruses, are used to deliver genetic material into eukaryotic cells for gene therapy and research applications.
Components: Transfer plasmid (gene of interest), packaging plasmid (viral proteins), envelope plasmid (host specificity).
Applications: Gene therapy, genome editing (e.g., CRISPR-Cas9), and functional genomics.
Applications of Recombinant DNA Technology
Gene Therapy
Gene therapy involves the delivery of therapeutic genes to correct genetic disorders. Genome editing tools like CRISPR-Cas9 enable precise modifications of the genome.
Example: Delivery of functional CFTR gene in cystic fibrosis.
Genome Editing: Addition, removal, or alteration of DNA at specific sites.
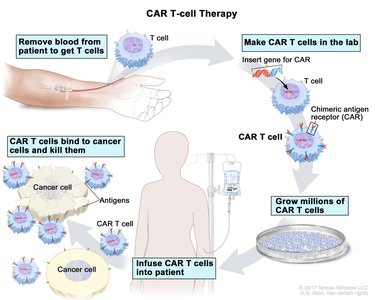
CAR T-cell Therapy
Chimeric Antigen Receptor (CAR) T-cell therapy is an advanced cancer treatment where a patient's T cells are genetically engineered to target and kill cancer cells.
Steps: T-cell collection, genetic engineering, expansion, and infusion into the patient.
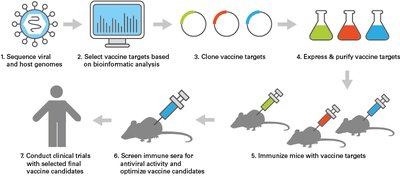
Vaccine Development
Recombinant DNA technology enables the development of vaccines by introducing antigens into cells using vectors, as seen in viral vector-based COVID-19 vaccines.
Functional Genomics
Vectors are used to study gene function through overexpression, downregulation (RNAi or CRISPR), and the creation of transgenic models.
Transgenic and Knockout Mouse Models
Transgenesis and Transgenic Mice
Transgenesis is the process of introducing foreign genetic material into an organism's genome, resulting in a gain of function. Transgenic mice are valuable for studying gene function, disease mechanisms, and drug responses.
Humanized Mice: Mice engineered to express human genes or cells for biomedical research.
Knockout Mice
Knockout mice are generated by disrupting a specific gene, resulting in a loss of function. This approach is crucial for understanding gene roles in physiology and disease.
Example: Leptin-knockout mice are used to study obesity and metabolism.
Methods for Generating Transgenic and Knockout Mice
There are several methods to introduce transgenes or create knockouts in mice:
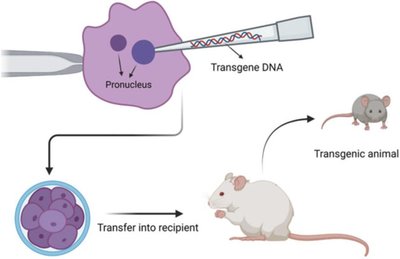
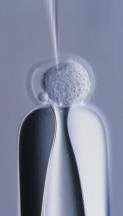
DNA Injection: Linearized DNA is injected into the pronucleus of a fertilized egg, which is then implanted into a surrogate mother.
Embryonic Stem Cell Approach: Modified ES cells are introduced into blastocysts to generate chimeric mice, which are bred to obtain homozygous knockouts.
Retroviral Approach: Retroviruses are used to deliver transgenes into embryos.
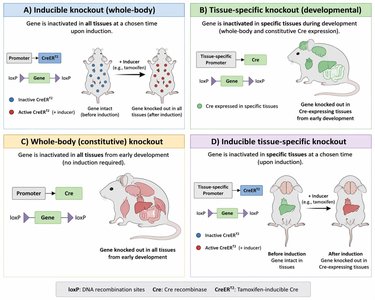
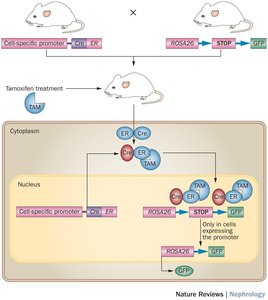
Conditional Knockout Mice: Cre-loxP System
The Cre-loxP system enables tissue-specific or inducible gene knockout. Cre recombinase recognizes loxP sites flanking a gene of interest and excises the gene in specific tissues or upon induction (e.g., tamoxifen treatment).
Tissue-Specific Knockout: Cre is expressed under a tissue-specific promoter.
Inducible Knockout: Cre is fused to a modified estrogen receptor, activated by tamoxifen for temporal control.
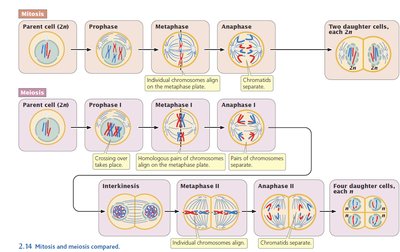
Mitosis, Meiosis, and Fertilization (Review)
Mitosis vs. Meiosis
Mitosis is the division of somatic cells, producing two genetically identical daughter cells. Meiosis is the division of germ cells, resulting in four genetically unique gametes with half the chromosome number.
Mitosis: Single division, no chromosome number reduction.
Meiosis: Two divisions, chromosome number halved, genetic recombination occurs.
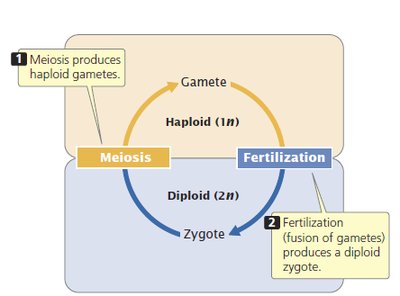
Fertilization and Early Embryogenesis
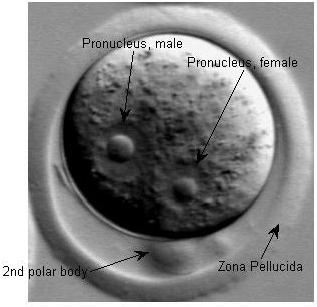
Fertilization restores the diploid chromosome number by combining genetic material from two gametes. The fertilized egg contains two pronuclei, which fuse to form the embryo's nucleus.
Pronuclei: Male pronucleus is typically larger due to chromatin decondensation.
Summary Table: General vs. Conditional Knockout Mice
Feature | General Knockout | Conditional Knockout |
|---|---|---|
Gene Inactivation | All tissues, early development | Specific tissues or time points |
Cre Expression | Not required | Required (tissue-specific or inducible) |
Temporal Control | No | Yes (inducible systems) |
Applications | Basic gene function | Study gene function in specific tissues or stages |
