 Back
BackTranslation and Post-Translational Modifications in Genetics
Study Guide - Smart Notes

Translation and Post-Translational Modifications
Overview of Translation
Translation is the process by which the genetic information encoded in messenger RNA (mRNA) is decoded by ribosomes and transfer RNAs (tRNAs) to synthesize polypeptides (proteins). This process is fundamental to gene expression and is highly conserved across all domains of life.
Key Components: mRNA, tRNA, ribosomes, amino acids, and various protein factors.
Stages: Initiation, elongation, and termination.
Genetic Code: The sequence of nucleotide triplets (codons) in mRNA determines the sequence of amino acids in the protein.

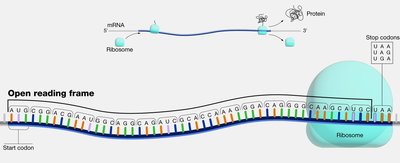
Open Reading Frame (ORF)
An open reading frame (ORF) is a continuous stretch of nucleotides in mRNA that begins with a start codon (usually AUG) and ends with a stop codon (UAA, UAG, or UGA). ORFs are the regions that can be translated into proteins.
Start Codon: Typically AUG, coding for methionine.
Stop Codons: UAA, UAG, UGA signal the end of translation.
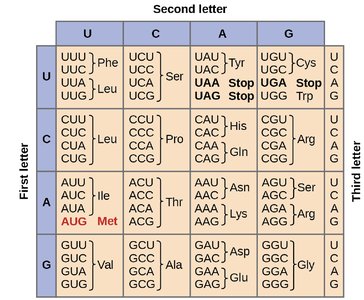
The Genetic Code and Codon Table
The genetic code is composed of 64 codons, each consisting of three nucleotides. It is degenerate, meaning that most amino acids are encoded by more than one codon. The first two bases of the codon are most critical for specifying the amino acid, while the third base is often less important (the "wobble" position).
Degeneracy: Multiple codons can code for the same amino acid.
Wobble Hypothesis: Flexibility in base pairing at the third codon position allows for fewer tRNAs to recognize multiple codons.
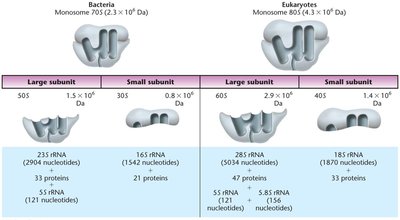
Translation Machinery: Ribosomes and tRNAs
Ribosomes are large ribonucleoprotein complexes composed of ribosomal RNA (rRNA) and proteins. They consist of a large and a small subunit. tRNAs are small, stable RNA molecules that serve as adapters, matching amino acids to their corresponding codons in the mRNA.
Prokaryotic Ribosomes: 70S (50S large + 30S small subunit)
Eukaryotic Ribosomes: 80S (60S large + 40S small subunit)
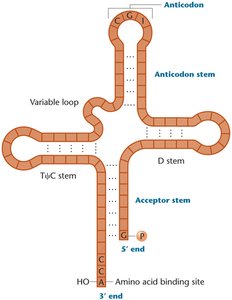
tRNA Structure: Cloverleaf secondary structure with an anticodon loop and an acceptor stem (3' CCA end for amino acid attachment).
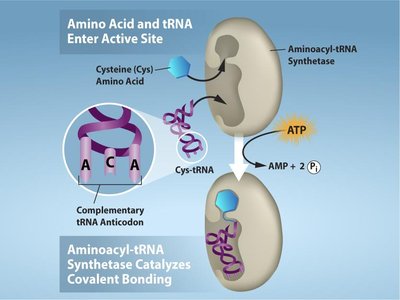
Charging of tRNAs
tRNAs are charged with their corresponding amino acids by enzymes called aminoacyl-tRNA synthetases. This process is highly specific and requires ATP.
20 Different Synthetases: One for each amino acid.
Reaction: Amino acid + tRNA + ATP → aminoacyl-tRNA + AMP + PPi
tRNA Modifications and Processing
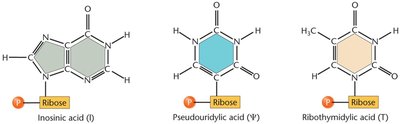
tRNAs undergo extensive post-transcriptional modifications, including base modifications, trimming of precursor sequences, addition of the CCA tail, and removal of introns. These modifications enhance tRNA stability and accuracy during translation.
Modified Bases: Inosine, pseudouridine, ribothymidine, etc.
Structural Processing: Removal of 5' leader and 3' trailer sequences, addition of CCA at the 3' end.
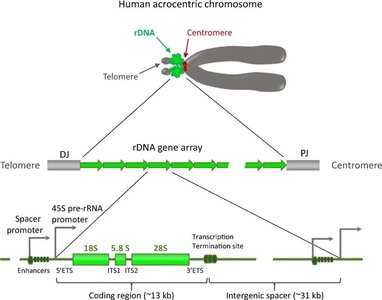
Ribosomal DNA (rDNA) Organization
rDNA refers to the DNA sequences that encode rRNA molecules. These genes are organized in tandem repeats and are found in clusters at specific chromosomal regions, such as the nucleolus organizer regions (NORs) in eukaryotes.
Human rDNA: Located on the short arms of acrocentric chromosomes (13, 14, 15, 21, 22).
Structure: Each repeat contains coding regions for 18S, 5.8S, and 28S rRNAs, separated by noncoding spacers.
Stages of Translation
Initiation (Prokaryotes)
In bacteria, translation initiation involves the small ribosomal subunit binding to the Shine–Dalgarno sequence on the mRNA, positioning the start codon (AUG) in the P site. The initiator tRNA carries N-formylmethionine (fMet) and three initiation factors (IF1, IF2, IF3) assist in complex formation.
Shine–Dalgarno Sequence: Ribosome-binding site upstream of AUG.
Initiator tRNA: Carries fMet in prokaryotes.
Elongation (Bacteria)
During elongation, aminoacyl-tRNAs enter the A site, peptide bonds are formed, and the ribosome translocates along the mRNA. Elongation factors (EF-Tu, EF-G) facilitate these steps.
Polysomes: Multiple ribosomes can translate a single mRNA simultaneously.
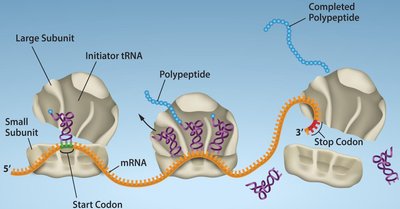
Termination
Termination occurs when a stop codon enters the A site. Release factors (RF1, RF2, RF3) recognize stop codons and promote the release of the newly synthesized polypeptide from the ribosome.
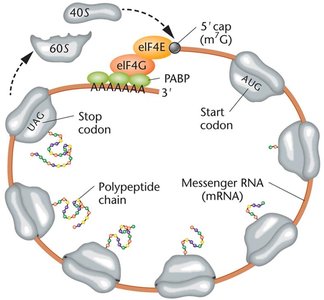
Translation in Eukaryotes
Eukaryotic translation is more complex, involving larger ribosomes, more initiation factors, and additional mRNA processing (5' cap, poly-A tail). The Kozak sequence helps ribosomes recognize the correct start codon.
5' Cap and Poly-A Tail: Essential for mRNA stability and translation initiation.
Kozak Sequence: Consensus sequence surrounding the start codon in eukaryotic mRNAs.
One-Gene–One-Enzyme and One-Gene–One-Polypeptide Hypotheses
Early genetic studies established that each gene encodes a specific enzyme (one-gene–one-enzyme hypothesis), later refined to one-gene–one-polypeptide as it became clear that not all proteins are enzymes and some proteins are composed of multiple polypeptides.
Beadle and Tatum Experiment: Used Neurospora crassa mutants to demonstrate the relationship between genes and enzymes.
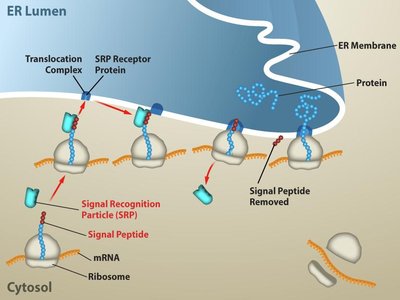
Protein Folding, Modification, and Targeting
After translation, polypeptides undergo folding and various modifications to become functional proteins. Signal sequences direct proteins to specific cellular compartments, and chaperones assist in proper folding.
Signal Sequences: Short amino-terminal sequences that direct proteins to organelles like the ER, mitochondria, or chloroplasts.
Chaperones: Proteins that assist in the folding of other proteins.
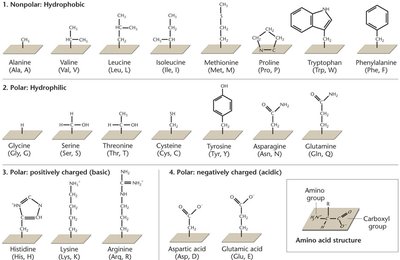
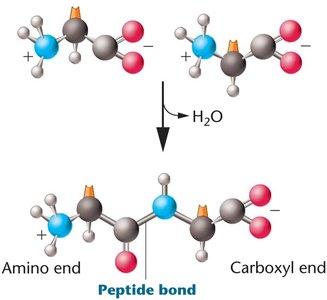
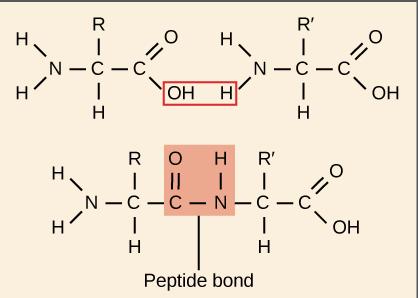
Amino Acids and Peptide Bonds
Amino acids are the building blocks of proteins, each with a central carbon, amino group, carboxyl group, and a variable R group. Peptide bonds form between the carboxyl group of one amino acid and the amino group of another via a dehydration reaction.
Types of Amino Acids: Nonpolar (hydrophobic), polar (hydrophilic), positively charged (basic), negatively charged (acidic).
Peptide Bond Formation:
Levels of Protein Structure
Proteins have four levels of structure:
Primary: Linear sequence of amino acids.
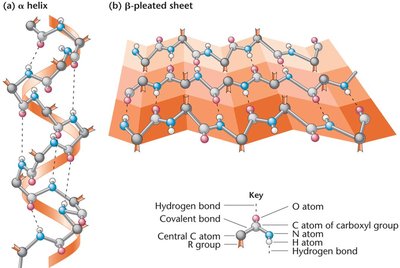
Secondary: Local folding into α-helices and β-pleated sheets.
Tertiary: Overall three-dimensional shape of a single polypeptide.
Quaternary: Association of multiple polypeptide chains.
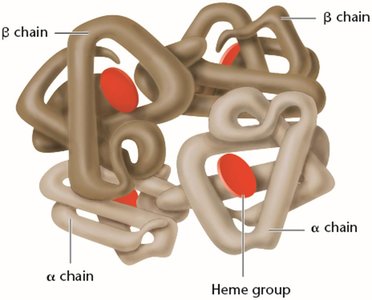
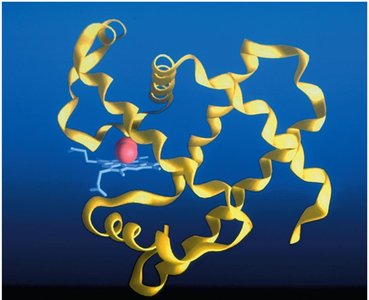
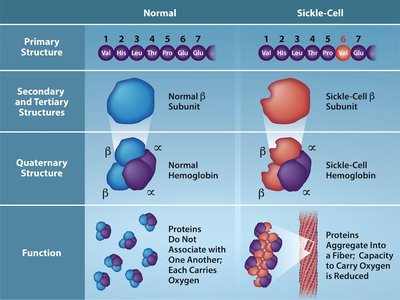
Mutations and Protein Function: Sickle-Cell Anemia
Mutations in the DNA sequence can alter the amino acid sequence of proteins, affecting their structure and function. Sickle-cell anemia is caused by a point mutation in the β-globin gene, resulting in the substitution of valine for glutamic acid at position 6, which leads to abnormal hemoglobin aggregation and sickle-shaped red blood cells.
Normal Hemoglobin (HbA): Glutamic acid at position 6.
Sickle Hemoglobin (HbS): Valine at position 6 due to a GAG → GTG mutation.
Diversity and Functions of Proteins
Proteins perform a wide variety of functions in cells, including structural support, catalysis, transport, signaling, and regulation of gene expression.
Examples: Collagen, keratin, actin, myosin, tubulin, immunoglobulins, enzymes, hormones, histones, transcription factors.
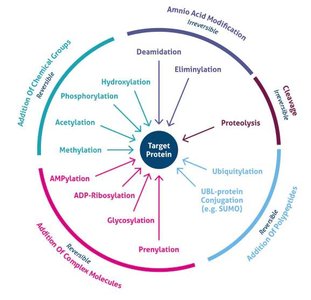
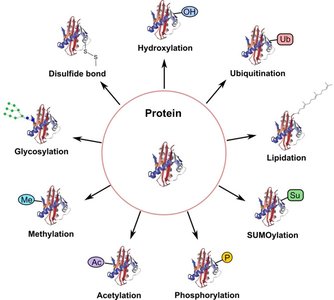
Post-Translational Modifications (PTMs)
After translation, proteins often undergo post-translational modifications that are essential for their function, stability, localization, and regulation. Common PTMs include phosphorylation, acetylation, methylation, glycosylation, ubiquitination, SUMOylation, lipidation, hydroxylation, and disulfide bond formation.
Modification | Main Function |
|---|---|
Phosphorylation | Regulates enzyme activity and cell signaling |
Acetylation | Controls gene expression and protein stability |
Methylation | Modulates protein interactions and gene regulation |
Glycosylation | Aids protein folding, stability, and cell recognition |
Ubiquitination | Tags proteins for degradation by the proteasome |
SUMOylation | Regulates protein localization and transcription |
Lipidation | Targets proteins to cell membranes |
Hydroxylation | Important for collagen stability and oxygen sensing |
Disulfide bonds | Stabilize protein structure |
Protein Domains and Functional Organization
Protein domains are distinct structural and functional units within a protein, typically 50–300 amino acids in length. Different domains confer specific functional capabilities, such as DNA binding, dimerization, ligand binding, or protein-protein interactions.
Transcription Factors: Modular proteins with DNA-binding and effector domains.
Protein-Protein Interactions: Induce conformational changes essential for function and signaling.
Regulation of Gene Expression
Gene expression is regulated at multiple levels, including chromatin structure, transcription, mRNA processing, translation, and protein degradation. Ubiquitination is a key post-translational modification that targets proteins for degradation by the proteasome, thereby controlling protein levels and activity in the cell.
Ubiquitin: Small protein that tags other proteins for degradation.
Proteasome: Organelle that degrades ubiquitinated proteins.
Summary: Translation and post-translational modifications are central to the flow of genetic information from DNA to functional proteins. Understanding these processes is essential for comprehending how genes determine phenotype and how mutations can lead to disease.
