 Back
BackAn Introduction to Organic Compounds: Nomenclature, Physical Properties, and Structure
Study Guide - Smart Notes

An Introduction to Organic Compounds
Overview of Organic Compounds and Their Classification
Organic chemistry is the study of carbon-containing compounds, particularly those involving carbon-carbon and carbon-hydrogen bonds. The classification, nomenclature, and physical properties of organic compounds are foundational for understanding their reactivity and applications in fields such as pharmaceuticals, materials science, and biochemistry.
Hydrocarbons and Alkanes
Definition and Structure of Alkanes
Alkanes are saturated hydrocarbons containing only single bonds between carbon atoms. Their general molecular formula is , where n is the number of carbon atoms. Alkanes can exist as straight-chain (normal) or branched isomers.
Hydrocarbons: Compounds composed solely of carbon and hydrogen.
Straight-chain alkanes: Alkanes with all carbons connected in a single, unbranched chain.
Branched alkanes: Alkanes with one or more branches (alkyl groups) attached to the main chain.
Example: Methane (), ethane (), propane (), and butane () are the simplest alkanes.
Constitutional Isomerism in Alkanes
Alkanes with four or more carbons can have constitutional isomers—compounds with the same molecular formula but different connectivity of atoms.
Isobutane is a branched isomer of butane ().
The number of possible isomers increases rapidly with the number of carbons.
Nomenclature of Alkanes and Alkyl Groups
Systematic (IUPAC) Nomenclature
The IUPAC system provides rules for naming organic compounds based on their structure:
Identify the longest continuous carbon chain (parent hydrocarbon).
Name and number substituents (alkyl groups) to give the lowest possible numbers.
List substituents in alphabetical order, using prefixes (di-, tri-, etc.) for multiples.
Example: 2-methylpropane (isobutane) and 2,2-dimethylpropane are isomers of pentane.
Alkyl Groups and Their Naming
Alkyl groups are derived from alkanes by removing one hydrogen atom. They are named by replacing the "-ane" ending with "-yl" (e.g., methyl, ethyl, propyl).
Primary, secondary, tertiary carbons: Classification depends on the number of other carbons attached to the carbon of interest.
Physical Properties of Alkanes and Related Compounds
Boiling and Melting Points
The boiling and melting points of alkanes increase with molecular weight due to greater van der Waals forces. Branching lowers boiling points by reducing surface contact between molecules.
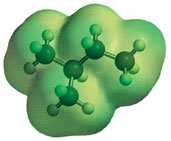
Solubility and Density
Alkanes are nonpolar and insoluble in water but soluble in nonpolar solvents. Their densities are less than that of water, causing them to float.
Functional Groups: Alkyl Halides, Alcohols, Ethers, and Amines
Alkyl Halides
Alkyl halides are compounds where a halogen (F, Cl, Br, I) replaces a hydrogen atom in an alkane. They are classified as primary, secondary, or tertiary based on the carbon to which the halogen is attached.
Alcohols
Alcohols contain an -OH group attached to a saturated carbon. They are classified as primary, secondary, or tertiary based on the carbon's connectivity.
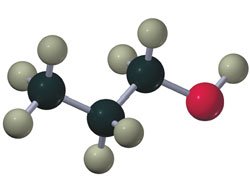
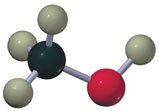
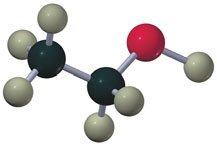
Ethers
Ethers have an oxygen atom bonded to two alkyl groups (R-O-R'). They can be symmetrical or unsymmetrical.
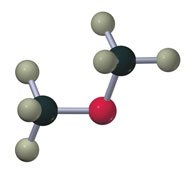
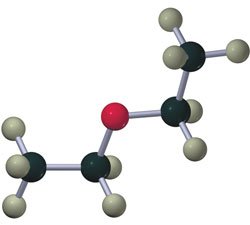
Amines
Amines are derivatives of ammonia (NH3) where one or more hydrogens are replaced by alkyl groups. They are classified as primary, secondary, or tertiary based on the number of alkyl groups attached to nitrogen.
Intermolecular Forces and Hydrogen Bonding
Types of Intermolecular Forces
Van der Waals forces: Weak, induced-dipole interactions present in all molecules, especially nonpolar ones like alkanes.
Dipole-dipole interactions: Stronger forces between polar molecules (e.g., alkyl halides, ethers).
Hydrogen bonds: Strong dipole-dipole interactions involving H attached to O, N, or F, as in alcohols and amines.
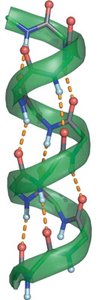
Conformational Analysis and Cycloalkanes
Conformers and Newman Projections
Rotation about single (sigma) bonds leads to different spatial arrangements called conformers. Newman projections are used to visualize these conformations.
Staggered conformer: More stable due to minimized electron repulsion.
Eclipsed conformer: Less stable due to increased electron repulsion.
Cycloalkanes and Ring Strain
Cycloalkanes are ring-shaped alkanes with the general formula . Smaller rings (e.g., cyclopropane, cyclobutane) have significant angle strain, while cyclohexane adopts a strain-free chair conformation.
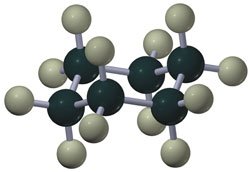
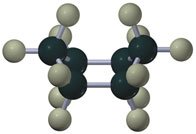
Substituted Cyclohexanes and Steric Interactions
Substituents on cyclohexane rings prefer equatorial positions to minimize steric (1,3-diaxial) interactions. The stability of different conformers can be predicted based on the size and position of substituents.
Biological and Practical Relevance
Octane Number and Fuel Quality
The octane number of gasoline measures its resistance to knocking in engines. Branched alkanes like 2,2,4-trimethylpentane have high octane numbers and are preferred in fuels.

Polysaccharides: Starch and Cellulose
Starch and cellulose are both polymers of glucose but differ in the type of glycosidic linkage, leading to different physical properties and biological roles.


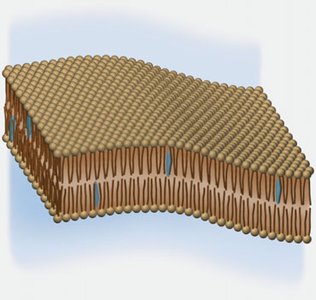
Cell Membranes and Lipid Bilayers
Cell membranes are composed of phospholipid bilayers, with nonpolar tails facing inward and polar heads facing outward, demonstrating the principle that "like dissolves like." Cholesterol, a rigid steroid, is an important membrane component.

Summary Table: Nomenclature of Key Functional Groups
Functional Group | Systematic Name | Common Name | Example |
|---|---|---|---|
Alkyl halide | Substituted alkane (e.g., bromomethane) | Alkyl halide (e.g., methyl bromide) | CH3Br |
Ether | Alkoxyalkane (e.g., methoxymethane) | Alkyl ether (e.g., dimethyl ether) | CH3OCH3 |
Alcohol | Alkanol (e.g., methanol) | Alkyl alcohol (e.g., methyl alcohol) | CH3OH |
Amine | Alkanamine (e.g., ethanamine) | Alkylamine (e.g., ethylamine) | CH3CH2NH2 |
Key Points to Remember
Systematic nomenclature allows for the unambiguous naming of organic compounds.
Physical properties such as boiling point, melting point, and solubility are determined by molecular structure and intermolecular forces.
Conformational analysis is essential for understanding the three-dimensional structure and reactivity of organic molecules.
Biological molecules and materials (e.g., cell membranes, starch, cellulose) are directly related to the principles of organic structure and bonding.
