 Back
BackOrganic Chemistry I: Structure, Nomenclature, and Functional Groups
Study Guide - Smart Notes

Bonding, Structure, and Reactivity
Atomic Structure and Orbitals
Organic chemistry is fundamentally the study of carbon-containing compounds, focusing on their structure, bonding, and reactivity. Atoms consist of a dense nucleus (protons and neutrons) surrounded by electrons in defined regions called orbitals. The arrangement of these electrons determines the chemical properties of the atom.
Atomic Orbitals: Regions of space with a high probability of finding an electron. The main types are s (spherical), p (dumbbell-shaped), d, and f orbitals.
Electron Configuration: The distribution of electrons among the orbitals, following the Aufbau principle, Pauli exclusion principle, and Hund’s rule.

Covalent Bonding and Hybridization
Covalent bonds form when atoms share electrons. Carbon’s ability to form four strong covalent bonds and to catenate (bond to itself) is central to organic chemistry. Hybridization explains the geometry of molecules:
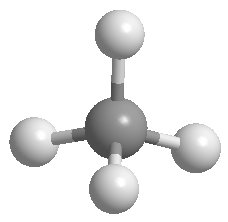
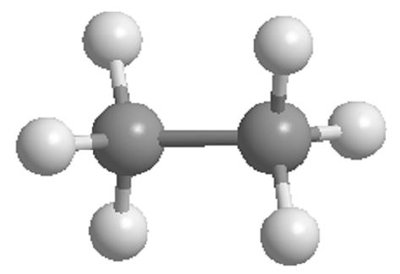
sp3 Hybridization: Tetrahedral geometry (e.g., methane, CH4), bond angles of 109.5°.
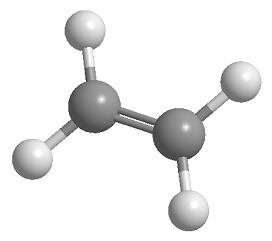
sp2 Hybridization: Trigonal planar geometry (e.g., ethene, C2H4), bond angles of 120°.
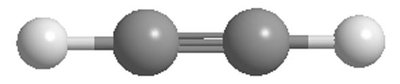
sp Hybridization: Linear geometry (e.g., acetylene, C2H2), bond angles of 180°.
Lewis Structures and Resonance
Lewis structures represent molecules showing all valence electrons. Multiple bonds and resonance structures are common in organic molecules, allowing for electron delocalization and increased stability.
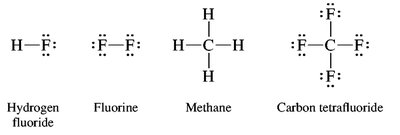
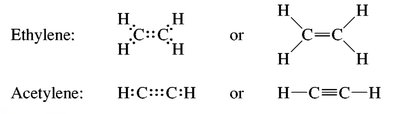
Functional Groups and Nomenclature
Functional Groups
Functional groups are specific groupings of atoms within molecules that determine the characteristic chemical reactions of those molecules. Recognizing and naming functional groups is essential for understanding organic reactivity.
Alcohols: –OH group
Alkyl halides: –X (X = F, Cl, Br, I)
Alkenes: C=C double bond
Alkynes: C≡C triple bond
Aromatic rings: Benzene and derivatives
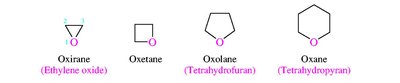
Ethers, amines, carbonyls, carboxylic acids, esters, amides, etc.
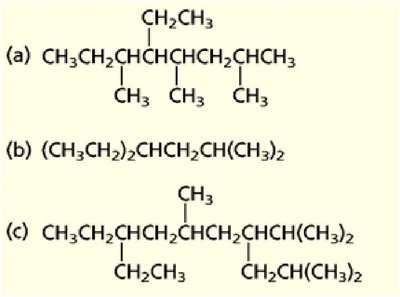
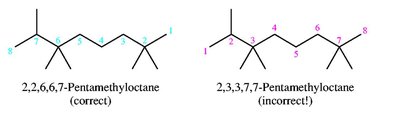
Nomenclature of Alkanes and Alkyl Groups
Alkanes are saturated hydrocarbons with the general formula CnH2n+2. The IUPAC system names alkanes based on the longest carbon chain and the nature and position of substituents.
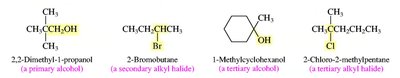
Primary, secondary, tertiary carbons: Classified by the number of other carbons attached.
Alkyl groups: Derived from alkanes by removing one hydrogen (e.g., methyl, ethyl, propyl, isopropyl, sec-butyl, tert-butyl).
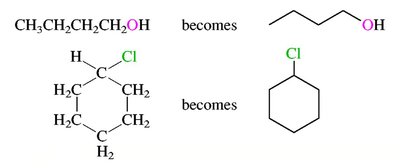
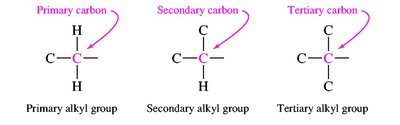
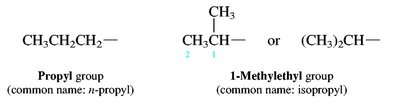
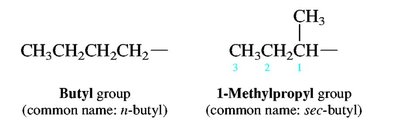
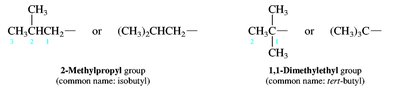
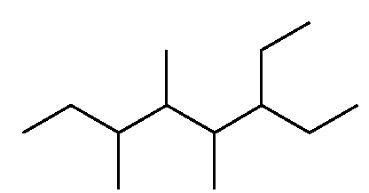
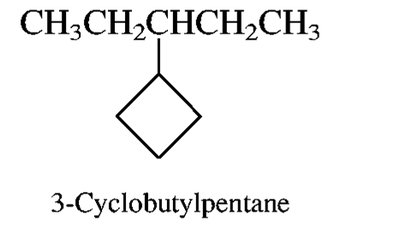
Branched and Cyclic Alkanes
Branched alkanes are named by identifying the longest chain and naming substituents as prefixes. Cycloalkanes are named by adding the prefix 'cyclo-' to the alkane name.
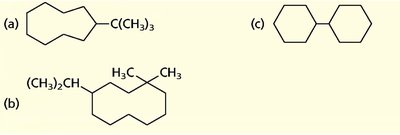
Polycyclic and Bicyclic Alkanes
Polycyclic alkanes contain more than one ring system. Bicyclic compounds are named using the bicyclo[x.y.z]alkane system, where x, y, and z are the number of carbons in each bridge.
![Bicyclo[3.2.1]octane structure](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/eb742655_image_20.png)
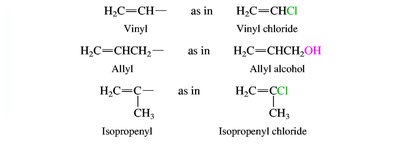
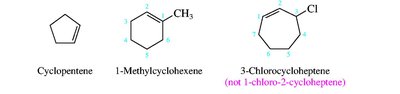
Nomenclature of Alkenes and Alkynes
Alkenes and alkynes are named by identifying the longest chain containing the multiple bond and numbering to give the lowest possible locant to the double or triple bond. Stereochemistry (cis/trans or E/Z) is specified for alkenes when necessary.
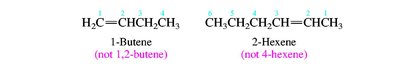

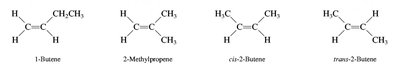
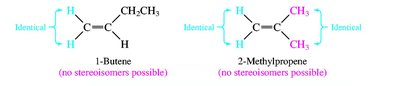
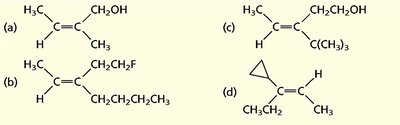
Nomenclature of Alcohols, Ethers, and Amines
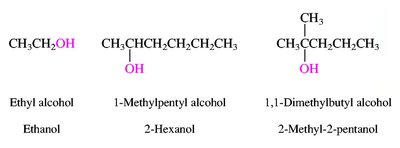
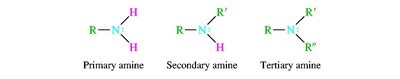
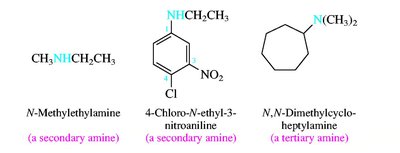
Alcohols are named by replacing the -e of the parent alkane with -ol and numbering to give the lowest possible number to the hydroxyl group. Ethers are named as alkoxyalkanes or by functional class names. Amines are classified as primary, secondary, or tertiary based on the number of alkyl groups attached to nitrogen.
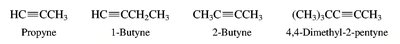

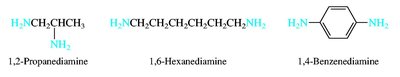
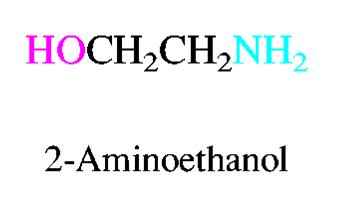
Nomenclature of Carbonyl Compounds
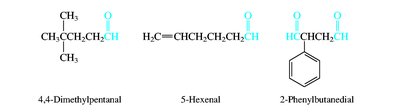
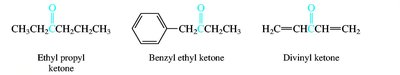
Aldehydes and ketones are named by replacing the -e of the parent alkane with -al (aldehydes) or -one (ketones). The carbonyl carbon is always given the lowest possible number. Common names are also widely used for simple compounds.
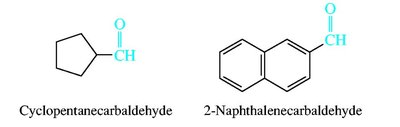
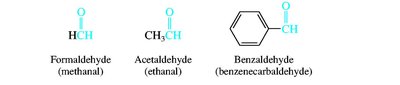
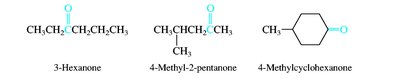
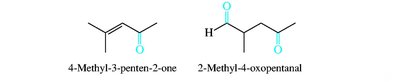
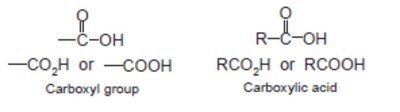
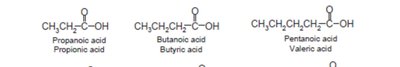
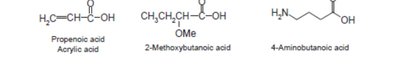
Nomenclature of Carboxylic Acids and Derivatives
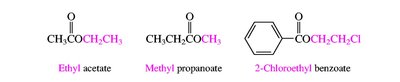
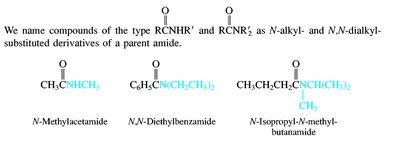
Carboxylic acids are named by replacing the -e of the parent alkane with -oic acid. Their derivatives (acid chlorides, anhydrides, esters, amides, nitriles) are named based on the parent acid, with appropriate suffixes and prefixes.
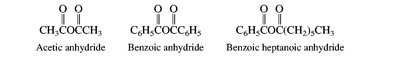


Additional info: This guide covers the foundational topics of organic chemistry, including atomic structure, bonding, hybridization, functional groups, and systematic nomenclature. Mastery of these concepts is essential for understanding organic reactivity and mechanisms in subsequent chapters.
