Textbook Question
For the reaction
b. Does entropy increase or decrease in this process?
1620
views

 Verified step by step guidance
Verified step by step guidance
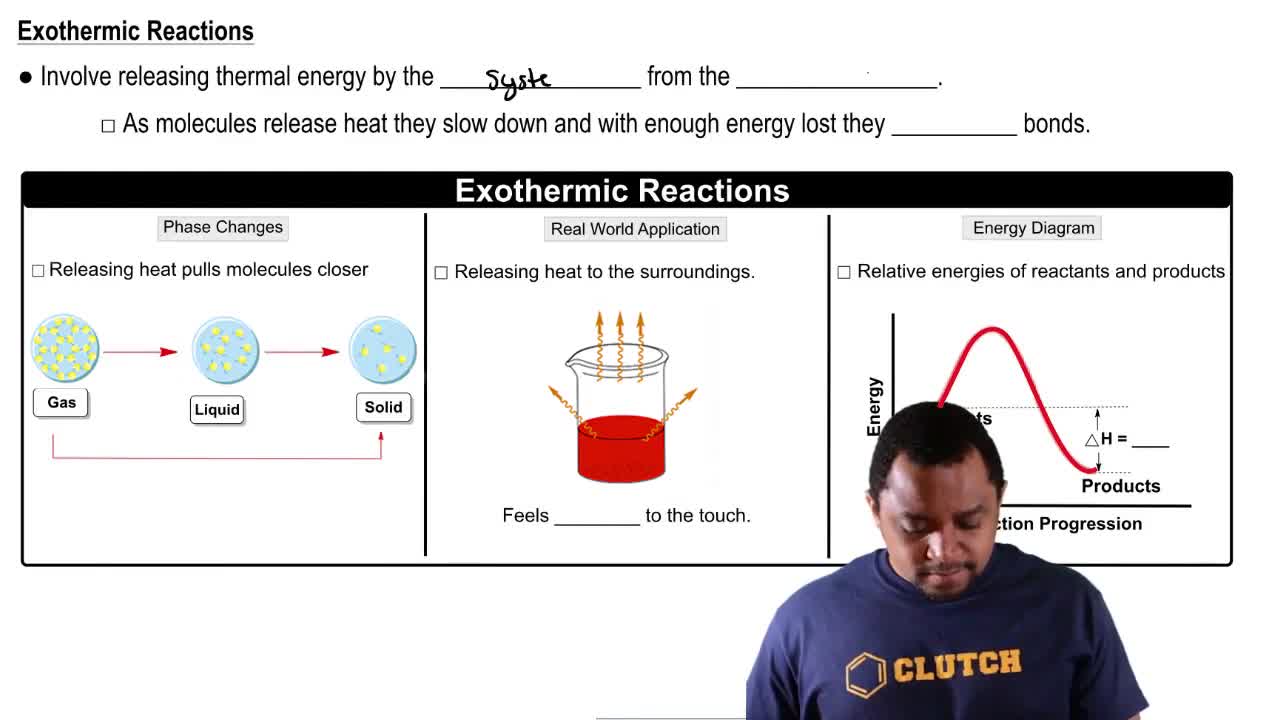

For the reaction
b. Does entropy increase or decrease in this process?
For each of the following processes, specify whether entropy increases or decreases. Explain each of your answers.
a. Assembling a jigsaw puzzle
What two factors affect the spontaneity of a reaction?
For the reaction 2 Hg(l) + O2(g) → 2 HgO(s), ∆H = –43 kcal/mol (–180 kJ/mol).
a. Does entropy increase or decrease in this process? Explain.
Which of the following processes results in an increase in entropy of the system?
a. A drop of ink spreading out when it is placed in water
b. Steam condensing into drops on windows
c. Constructing a building from loose bricks
Under what conditions might a reaction be endothermic but exergonic? Explain.