An argon ion laser puts out 5.0 W of continuous power at a wavelength of 532 nm. The diameter of the laser beam is 5.5 mm. If the laser is pointed toward a pinhole with a diameter of 1.2 mm, how many photons travel through the pinhole per second? Assume that the light intensity is equally distributed throughout the entire cross-sectional area of the beam. (1 W = 1 J/s)
Ch.7 - Quantum-Mechanical Model of the Atom

Chapter 7, Problem 78
An X-ray photon of wavelength 0.989 nm strikes a surface. The emitted electron has a kinetic energy of 969 eV. What is the binding energy of the electron in kJ/mol? [KE = 1/2 mv2; 1 electron volt (eV) = 1.602×10–19 J]
 Verified step by step guidance
Verified step by step guidance1
Convert the wavelength of the X-ray photon from nanometers to meters by using the conversion factor: 1 nm = 1 x 10^{-9} m.
Calculate the energy of the X-ray photon using the formula E = \frac{hc}{\lambda}, where h is Planck's constant (6.626 x 10^{-34} J\cdot s) and c is the speed of light (3.00 x 10^8 m/s).
Convert the kinetic energy of the emitted electron from electron volts (eV) to joules (J) using the conversion factor: 1 eV = 1.602 x 10^{-19} J.
Determine the binding energy of the electron by subtracting the kinetic energy (in joules) from the energy of the X-ray photon (in joules).
Convert the binding energy from joules to kilojoules per mole by using Avogadro's number (6.022 x 10^{23} mol^{-1}) and the conversion factor: 1 kJ = 1000 J.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Was this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Photoelectric Effect
The photoelectric effect describes the phenomenon where electrons are emitted from a material when it absorbs light or electromagnetic radiation, such as X-rays. The energy of the incoming photon must exceed the binding energy of the electrons in the material for them to be released. The kinetic energy of the emitted electrons can be calculated by subtracting the binding energy from the energy of the incoming photon.
Recommended video:
Guided course
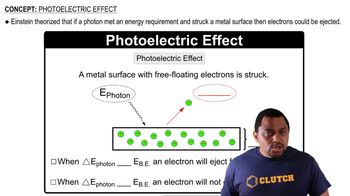
Photoelectric Effect
Energy of a Photon
The energy of a photon is inversely related to its wavelength and can be calculated using the equation E = hc/λ, where E is energy, h is Planck's constant, c is the speed of light, and λ is the wavelength. In this question, the wavelength of the X-ray photon is given, allowing us to determine its energy, which is crucial for calculating the binding energy of the emitted electron.
Recommended video:
Guided course

Photon Energy Formulas
Conversion of Energy Units
In this problem, it is necessary to convert energy units from electron volts (eV) to kilojoules per mole (kJ/mol). The conversion factor is 1 eV = 1.602 x 10^-19 J, and to convert joules to kilojoules, one divides by 1000. Additionally, to convert from energy per particle to energy per mole, Avogadro's number (6.022 x 10^23 mol^-1) is used, which is essential for expressing the binding energy in the desired units.
Recommended video:
Guided course

Conversion Factors
Related Practice
Textbook Question
1008
views
Textbook Question
Ionization involves completely removing an electron from an atom. How much energy is required to ionize a hydrogen atom in its ground (or lowest energy) state? What wavelength of light contains enough energy in a single photon to ionize a hydrogen atom?
1801
views
1
comments
Textbook Question
The energy required to ionize sodium is 496 kJ/mol. What minimum frequency of light is required to ionize sodium?
1739
views
1
rank
Textbook Question
Suppose that in an alternate universe, the possible values of l are the integer values from 0 to n (instead of 0 to n - 1). Assuming no other differences between this imaginary universe and ours, how many orbitals would exist in each level? a. n = 1 b. n = 2 c. n = 3
1073
views
