12. Molecular Shapes & Valence Bond Theory
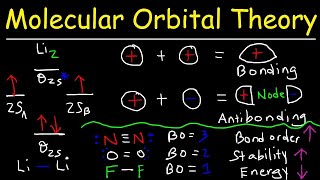
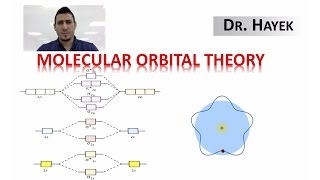
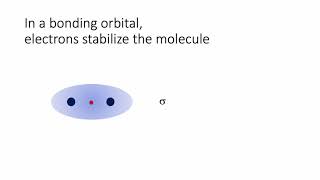
Molecular Orbital Theory

Get help from an AI Tutor
Ask a question to get started.
Problem 74c
Textbook Question
Textbook QuestionSketch the bonding and antibonding molecular orbitals that result from linear combinations of the 2pz atomic orbitals in a homonuclear diatomic molecule. (The 2pz orbitals are those whose lobes are oriented perpendicular to the bonding axis.) How do these molecular orbitals differ from those obtained from linear combinations of the 2py atomic orbitals? (The 2py orbitals are also oriented perpendicular to the bonding axis, but also perpendicular to the 2pz orbitals.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
582
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos