The value of Ksp for Cd(OH)2 is 2.5 × 10–14. (b) The solubility of Cd(OH)2 can be increased through formation of the complex ion CdBr42- (Kf = 5 × 103). If solid Cd(OH)2 is added to a NaBr solution, what is the initial concentration of NaBr needed to increase the molar solubility of Cd(OH)2 to 1.0 × 10-3 mol/L?
Ch.17 - Additional Aspects of Aqueous Equilibria

Brown14th EditionChemistry: The Central ScienceISBN: 9780134414232Not the one you use?Change textbook
Chapter 17, Problem 111b
(b) As the acid is titrated, the pH of the solution after the addition of 11.05 mL of the base is 4.89. What is the Ka for the acid?
 Verified step by step guidance
Verified step by step guidance1
Identify the type of titration: Since the pH is given after adding a base to an acid, this is an acid-base titration.
Use the Henderson-Hasselbalch equation: \( \text{pH} = \text{pK}_a + \log \left( \frac{[\text{A}^-]}{[\text{HA}]} \right) \), where \([\text{A}^-]\) is the concentration of the conjugate base and \([\text{HA}]\) is the concentration of the acid.
Rearrange the equation to solve for \( \text{pK}_a \): \( \text{pK}_a = \text{pH} - \log \left( \frac{[\text{A}^-]}{[\text{HA}]} \right) \).
Convert \( \text{pK}_a \) to \( K_a \) using the relationship \( K_a = 10^{-\text{pK}_a} \).
Determine the concentrations of \([\text{A}^-]\) and \([\text{HA}]\) using the initial concentrations and volumes of the acid and base to find \( K_a \).

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
1mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Acid-Base Titration
An acid-base titration is a quantitative analytical method used to determine the concentration of an acid or base in a solution. During the titration, a base is gradually added to an acid (or vice versa) until the reaction reaches the equivalence point, where the amount of acid equals the amount of base. The pH of the solution changes throughout the process, and specific pH values can indicate the strength of the acid or base involved.
Recommended video:
Guided course
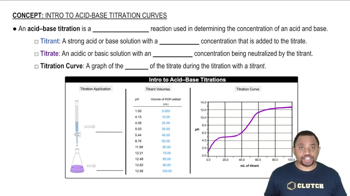
Acid-Base Titration
pH and pKa Relationship
The pH of a solution is a measure of its acidity or basicity, defined as the negative logarithm of the hydrogen ion concentration. The pKa is the negative logarithm of the acid dissociation constant (Ka) and indicates the strength of an acid; lower pKa values correspond to stronger acids. The Henderson-Hasselbalch equation relates pH, pKa, and the concentrations of the acid and its conjugate base, allowing for the calculation of Ka when pH and concentrations are known.
Recommended video:
Guided course
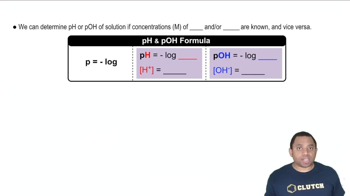
pH and pOH Calculations
Acid Dissociation Constant (Ka)
The acid dissociation constant (Ka) quantifies the strength of an acid in solution, representing the equilibrium constant for the dissociation of the acid into its ions. A higher Ka value indicates a stronger acid that dissociates more completely in solution. The Ka can be calculated using the concentrations of the products and reactants at equilibrium, which is essential for determining the strength of the acid in the titration scenario presented.
Recommended video:
Guided course

Characteristics of Ka and Kb
Related Practice
Textbook Question
405
views
Textbook Question
(a) Write the net ionic equation for the reaction that occurs when a solution of hydrochloric acid (HCl) is mixed with a solution of sodium formate 1NaCHO22.
1573
views
Textbook Question
(a) A 0.1044-g sample of an unknown monoprotic acid requires 22.10 mL of 0.0500 M NaOH to reach the end point. What is the molar mass of the unknown?
1774
views
Textbook Question
What is the pH at 25 C of water saturated with CO2 at a partial pressure of 1.10 atm? The Henry's law constant for CO2 at 25 C is 3.1 * 10-2 mol>L@atm.
840
views
