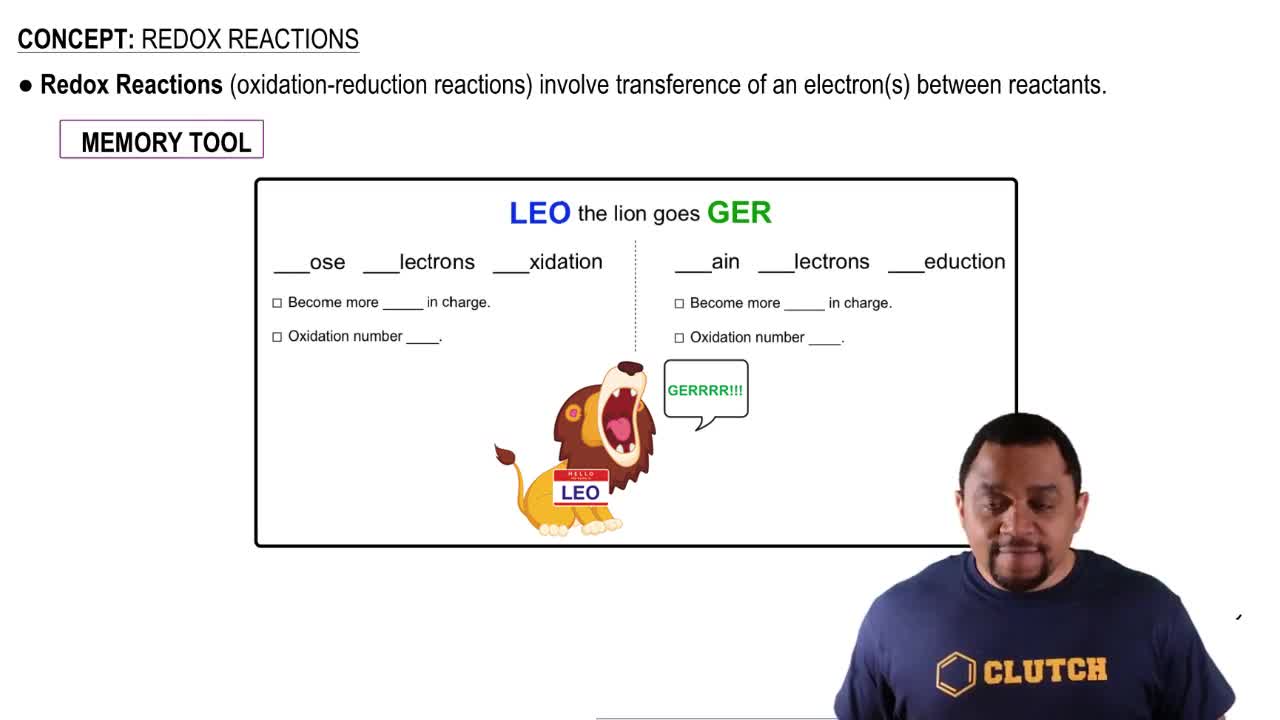
Textbook Question
Electrostatic potential maps of acetic acid (CH3CO2H) and ethyl alcohol (CH3CH2OH) are shown. Identify the most acidic hydrogen in each, and tell which of the two is likely to be the stronger acid.
<IMAGE>
2228
views

 Verified step by step guidance
Verified step by step guidance

Electrostatic potential maps of acetic acid (CH3CO2H) and ethyl alcohol (CH3CH2OH) are shown. Identify the most acidic hydrogen in each, and tell which of the two is likely to be the stronger acid.
<IMAGE>
Show how ethylamine (C2H5NH2) reacts with hydrochloric acid to form an ethylammonium salt.
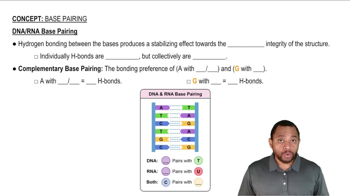
From this electrostatic potential map of the amino acid alanine, identify the most acidic hydrogens in the molecule:
<IMAGE>