Textbook Question
Can one of the possible molecular formulas contain a nitrogen atom?
1095
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy
Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy Problem 11
Problem 11 Verified step by step guidance
Verified step by step guidance
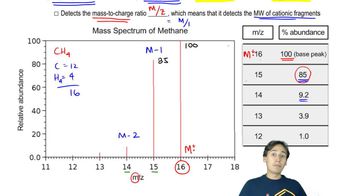

Can one of the possible molecular formulas contain a nitrogen atom?
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown below. Match each compound with its spectrum.
<IMAGE>
Identify the hydrocarbon that has a molecular ion with an m/z value of 128, a base peak with an m/z value of 43, and significant peaks with m/z values of 57, 71, and 85.
Primary alcohols have a strong peak at m/z = 31.
a. What fragment is responsible for this peak?
Predict the relative intensities of the molecular ion peak, the M+2 peak, and the M+4 peak for a compound that contains two bromine atoms.
Using curved arrows, show the principal fragments you would expect to see in the mass spectrum of each of the following compounds:
b.