Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
(c)

 Verified step by step guidance
Verified step by step guidance



Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
(c)
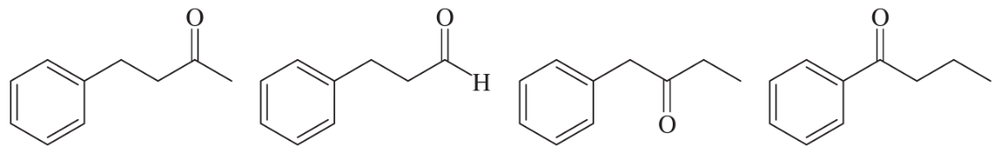
Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
(a)
One of these reacts with dilute aqueous acid and the other does not. Give a mechanism for the one that reacts, and show why this mechanism does not work for the other one.
(b)
Which of the following compounds would give a positive Tollens test? (Remember that the Tollens test involves mild basic aqueous conditions.)
(d) CH3CH2CH2CH2CH(OH)OCH3
(e) CH3CH2CH2CH2CH(OCH3)2
(f)
One of these reacts with dilute aqueous acid and the other does not. Give a mechanism for the one that reacts, and show why this mechanism does not work for the other one.
(a)
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K.