Textbook Question
How can a nucleus emit an electron during β decay when there are no electrons present in the nucleus to begin with?
2197
views

 Verified step by step guidance
Verified step by step guidance

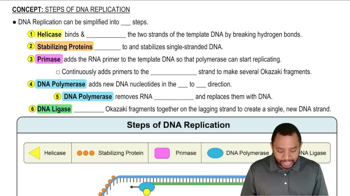
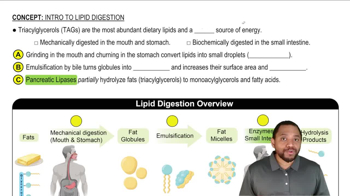
How can a nucleus emit an electron during β decay when there are no electrons present in the nucleus to begin with?
How does nuclear fission differ from normal radioactive decay?
Identify and write the symbol for each of the five nuclides in the decay series shown in Problem 11.25.
Identify the starting radioisotopes needed to balance each of these nuclear reactions:
a. ? + 42He → 11349In
b. ? + 42He → 137N + 10n