Textbook Question
How could you make the zwitterion of aspartic acid more soluble in water?
838
views

 Verified step by step guidance
Verified step by step guidance


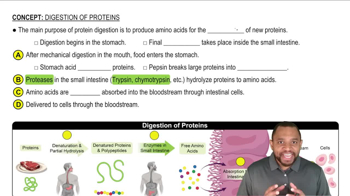
How could you make the zwitterion of aspartic acid more soluble in water?
Which of the following forms of aspartic acid would you expect to predominate at low pH, neutral pH, and high pH?
a.
b.
c.
Use the three-letter abbreviations to name all tripeptides that contain valine, methionine, and leucine.
Which form of aspartic acid in Problem 18.54 is the zwitterion? What is the pI for the zwitterion?
a.
b.
c.
Write structural formulas for the two dipeptides that contain leucine and aspartate.
At neutral pH, which of the following amino acids has a net positive charge, which has a net negative charge, and which is neutral? (Hint: Draw the various charged forms of each amino acid before deciding.)
a. Asparagine
b. Lysine
c. Proline