A local weather station reports the barometric pressure as 29.5 inHg (inches of Hg). Convert this pressure to torr and to atm.

Would you expect the boiling points to increase or decrease in the following series? Explain.
a. Kr, Ar, Ne
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
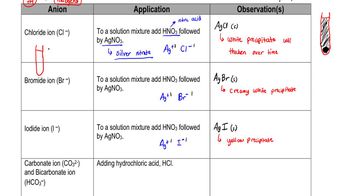
Noble Gases
Atomic Size and Boiling Point

Trends in the Periodic Table

Determine the percent composition of air in the lungs from the following composition in partial pressures: PN2= 573 mmHg, PO2 = 100 mmHg, PCO2 = 40 mmHg, and PH2O = 47 mmHg; all at 37 °C and 1 atm pressure.
The change of state from liquid H2O to gaseous H2O has ∆H = +9.72 kcal/mol(+40.7 kJ/mol) and ∆S = -26.1 cal/(mol • K) [-109 J/(mol •K)].
a. Is the change from liquid to gaseous H2O favored or unfavored by ∆H? By ∆S?
Compare the ∆Hvap values for water, isopropyl alcohol, ether, and ammonia, and order them from lowest to highest. Explain the rank order based on intermolecular attractive forces.
The change of state from liquid H2O to gaseous H2O has ∆H = +9.72 kcal/mol(+40.7 kJ/mol) and ∆S = -26.1 cal/(mol • K) [-109 J/(mol •K)].
b. What are the values of ∆H and ∆S (in kcal/mol and kJ/mol) for the change from gaseous to liquid H2O?
