Textbook Question
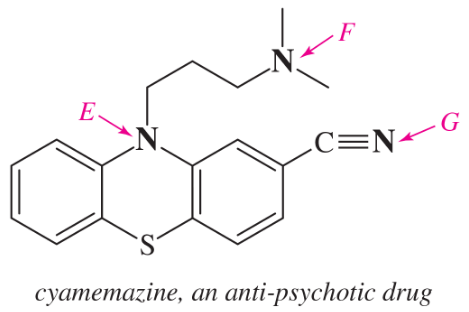
Within each structure, rank the indicated nitrogens by increasing basicity.
(a)
(b)
1213
views

 Verified step by step guidance
Verified step by step guidance


Within each structure, rank the indicated nitrogens by increasing basicity.
(a)
(b)
Rank the amines in each set in order of increasing basicity.
(e)
Within each structure, rank the indicated nitrogens by increasing basicity.
(d)
(e)
Within each structure, rank the indicated nitrogens by increasing basicity.
(f)
In each pair of compounds, select the stronger base, and explain your choice.
(a) HOCH2CH2NH2 or CH3CH2NH2
(b) PhNH2 or PhCH2NH2
Rank the amines in each set in order of increasing basicity.
(d)