Ammonia, NH3, is very soluble in water (51.8 g/L at 20 °C and 760 mmHg).
a. Show how NH3 can hydrogen bond to water.

 Verified step by step guidance
Verified step by step guidance


Ammonia, NH3, is very soluble in water (51.8 g/L at 20 °C and 760 mmHg).
a. Show how NH3 can hydrogen bond to water.
Cobalt(II) chloride, a blue solid, can absorb water from the air to form cobalt(II) chloride hexahydrate, a pink solid. The equilibrium is so sensitive to moisture in the air that CoCl2 is used as a humidity indicator.
a. Write a balanced equation for the equilibrium. Be sure to include water as a reactant to produce the hexahydrate.
How many milliliters of 0.150 M BaCl2 are needed to react completely with 35.0 mL of 0.200 M Na2SO4? How many grams of BaSO4 will be formed?
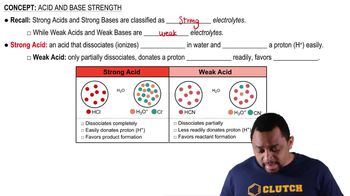
Many compounds are only partially dissociated into ions in aqueous solution. Trichloroacetic acid (CCl3CO2H), for instance, is partially dissociated in water according to the equation
CCl3CO2H(aq) → H+(aq) + CCl3CO2⁻ aq)
For a solution prepared by dissolving 1.00 mol of trichloroacetic acid in 1.00 kg of water, 36.0% of the trichloroacetic acid dissociates to form H+ and CCl3CO2⁻ ions.
b. What is the freezing point of this solution? (The freezing point of 1 kg of water is lowered 1.86 °C for each mole of solute particles.)
Hyperbaric chambers, which provide high pressures (up to 6 atm) of either air or pure oxygen, are used to treat a variety of conditions, ranging from decompression sickness in deep-sea divers to carbon monoxide poisoning. Look up the solubility of O2, N2, CO, and CO2 in water at standard temperature and pressure (1 atm, 25 °C).
a. Explain the trends in relative solubility for these gases.
Hyperbaric chambers, which provide high pressures (up to 6 atm) of either air or pure oxygen, are used to treat a variety of conditions, ranging from decompression sickness in deep-sea divers to carbon monoxide poisoning. Look up the solubility of O2, N2, CO, and CO2 in water at standard temperature and pressure (1 atm, 25 °C).
b. Explain how elevated pressures in a hyperbaric chamber be used to treat decompression sickness (excess N2 in blood) and carbon monoxide poisoning.