Textbook Question
Indicate whether each statement is true or false. (b) Exothermic reactions are faster than endothermic reactions.
868
views

 Verified step by step guidance
Verified step by step guidance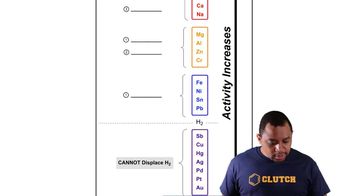


Indicate whether each statement is true or false. (b) Exothermic reactions are faster than endothermic reactions.
Indicate whether each statement is true or false. (a) If you measure the rate constant for a reaction at different temperatures, you can calculate the overall enthalpy change for the reaction.
Indicate whether each statement is true or false. (c) Increasing the reaction temperature increases the fraction of successful collisions between reactants.