Textbook Question
Determine the number of moles (of molecules or formula units) in each sample. a. 55.98 g CF2Cl2
895
views

 Verified step by step guidance
Verified step by step guidance


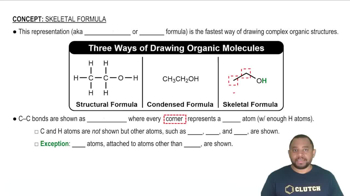
Determine the number of moles (of molecules or formula units) in each sample. a. 55.98 g CF2Cl2
Determine the number of moles (of molecules or formula units) in each sample. c. 0.1187 g C8H18
Refer to the nomenclature flowchart (Figure 3.11) to name each compound. a. XeO3 b. KClO
Calculate the formula mass for each compound. c. H2SO4
Determine the number of moles (of molecules or formula units) in each sample. b. 23.6 kg Fe(NO3)2
Refer to the nomenclature flowchart (Figure 3.11) to name each compound. c. CoSO4