The following cell reactions occur spontaneously: (a) Arrange the following reduction half-reactions in order of decreasing tendency to occur:
Ch.19 - Electrochemistry

Chapter 19, Problem 80a
Use the data in Appendix D to predict whether the following reactions can occur under standard-state conditions. (a) Oxidation of Sn2+(aq) by Br2(aq)
 Verified step by step guidance
Verified step by step guidance1
Identify the half-reactions involved in the oxidation of \( \text{Sn}^{2+} \) by \( \text{Br}_2 \).
Write the reduction half-reaction for \( \text{Br}_2 \) and the oxidation half-reaction for \( \text{Sn}^{2+} \).
Use Appendix D to find the standard reduction potentials \( E^0 \) for each half-reaction.
Calculate the standard cell potential \( E^0_{\text{cell}} \) by subtracting the oxidation potential from the reduction potential.
Determine if the reaction is spontaneous under standard conditions by checking if \( E^0_{\text{cell}} > 0 \).

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
3mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Standard-State Conditions
Standard-state conditions refer to a set of specific conditions used to measure the properties of substances. These conditions typically include a temperature of 25°C (298 K), a pressure of 1 atm, and concentrations of 1 M for solutions. Understanding these conditions is crucial for predicting the feasibility of chemical reactions, as they provide a baseline for comparing thermodynamic data.
Recommended video:
Guided course

Standard Reduction Potentials
Oxidation-Reduction Reactions
Oxidation-reduction (redox) reactions involve the transfer of electrons between species, resulting in changes in oxidation states. In the given reaction, Sn<sup>2+</sup> is oxidized to Sn<sup>4+</sup>, while Br<sub>2</sub> is reduced to Br<sup>-</sup>. Identifying the oxidizing and reducing agents is essential for predicting whether the reaction can occur spontaneously under standard-state conditions.
Recommended video:
Guided course
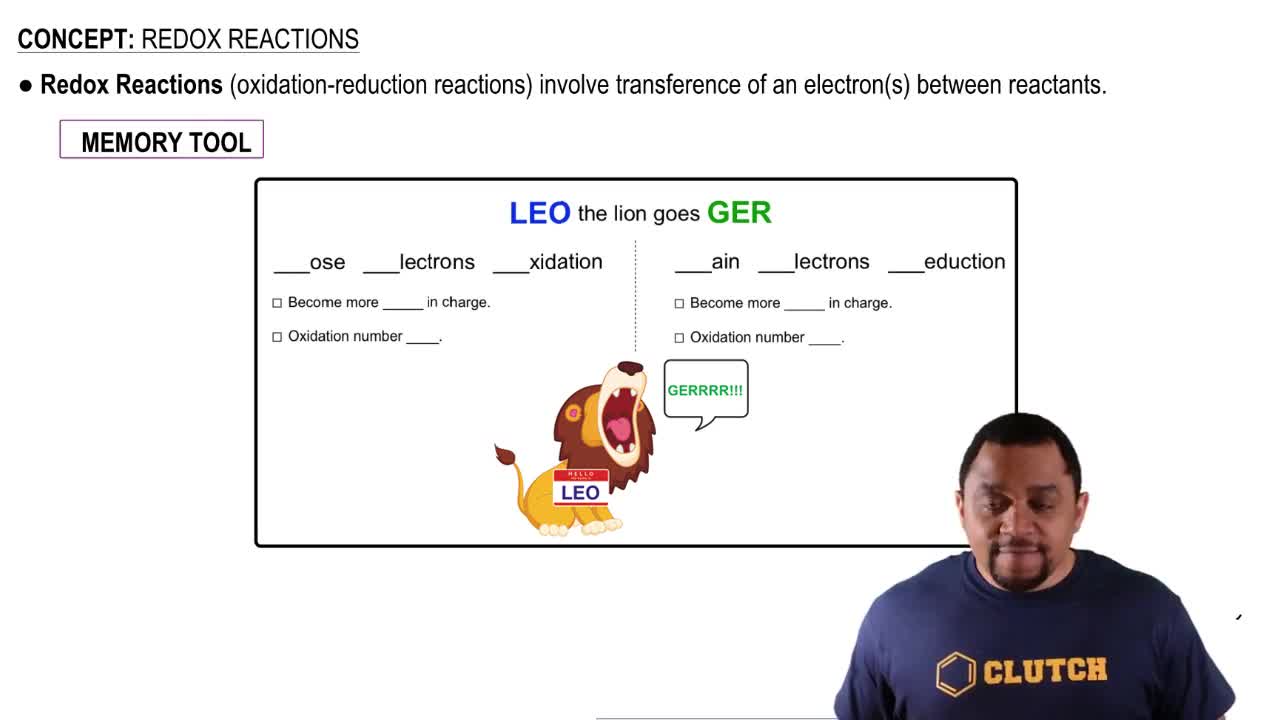
Oxidation and Reduction Reactions
Electrochemical Cell Potential
The electrochemical cell potential, or standard cell potential (E°), is a measure of the driving force behind a redox reaction. It can be calculated using standard reduction potentials from tables. A positive E° indicates that the reaction is spontaneous under standard-state conditions, while a negative E° suggests it is non-spontaneous. This concept is vital for predicting the feasibility of the oxidation of Sn<sup>2+</sup> by Br<sub>2</sub>.
Recommended video:
Guided course

Electrochemical Cells
Related Practice
Textbook Question
370
views
Textbook Question
The following cell reactions occur spontaneously: (b) Which of these substances (A,A+,B,B+,C,C+) is the strongest oxidizing agent? Which is the strongest reducing agent?
573
views
Textbook Question
Use the data in Appendix D to predict whether the following reactions can occur under standard-state conditions. (d) Reduction of I2(s) by H2SO3(aq)
391
views
Textbook Question
The following cell reactions occur spontaneously: (c) Which of the three cell reactions delivers the highest voltage?
315
views
Textbook Question
Use the data in Appendix D to predict whether the following reactions can occur under standard-state conditions. (b) Reduction of Ni2+(aq) by Sn2+(aq)
299
views
Textbook Question
Use the data in Appendix D to predict whether the following reactions can occur under standard-state conditions. (c) Oxidation of Ag(s) by Pb2+(aq)
341
views
