Textbook Question
(a) An AB2 molecule is linear. How many nonbonding electron pairs are around the A atom from this information?
880
views

 Verified step by step guidance
Verified step by step guidance
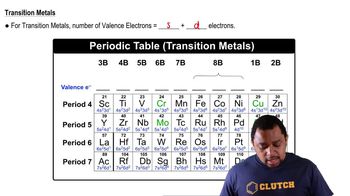

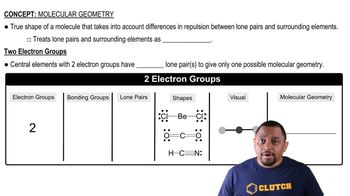
(a) An AB2 molecule is linear. How many nonbonding electron pairs are around the A atom from this information?
For each of these contour representations of molecular orbitals, identify (c) whether the MO is bonding or antibonding (i)
Describe the bond angles to be found in each of the following molecular structures: (a) trigonal planar, (b) tetrahedral, (c) octahedral, (d) linear.
(a) Boron trichloride 1BCl32 and the carbonate ion 1CO3 2- 2 are both described as trigonal. What does this indicate about their bond angles?
(b) The PCl3 molecule is trigonal pyramidal, while ICl3 is T-shaped. Which of these molecules is flat?
For each of these contour representations of molecular orbitals, identify (b) the type of MO (s or p) (i)