Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfur tetrafluoride monoxide (OSF4) according to the following unbalanced reaction: SF4(g) + O2(g) → OSF4(g) The O atom and the four F atoms in OSF4 are bonded to a central S atom. (b) Write a Lewis structure of OSF4 in which the formal charges of all atoms are zero.
Ch.9 - Molecular Geometry and Bonding Theories

Brown14th EditionChemistry: The Central ScienceISBN: 9780134414232Not the one you use?Change textbook
Chapter 9, Problem 115
The phosphorus trihalides 1PX32 show the following variation in the bond angle X¬P¬X: PF3, 96.3°; PCl3, 100.3°; PBr3, 101.0°; PI3, 102.0°. The trend is generally attributed to the change in the electronegativity of the halogen. (b) What is the general trend in the X¬P¬X angle as the halide electronegativity increases?
 Verified step by step guidance
Verified step by step guidance1
Identify the electronegativities of the halogens F, Cl, Br, and I. Recall that electronegativity generally decreases down the periodic table.
Analyze the given bond angles for each phosphorus trihalide: PF3 (96.3°), PCl3 (100.3°), PBr3 (101.0°), PI3 (102.0°).
Observe the relationship between the decreasing electronegativity of the halogens and the increasing bond angle in the phosphorus trihalides.
Consider the effect of electronegativity on electron density around the phosphorus atom and how it influences the repulsion between bonding pairs of electrons.
Conclude the general trend: as the electronegativity of the halide X in PX3 decreases, the X-P-X bond angle increases.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
2mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Electronegativity
Electronegativity is a measure of an atom's ability to attract and hold onto electrons in a chemical bond. In the context of phosphorus trihalides, the electronegativity of the halogen atoms (F, Cl, Br, I) influences the bond angles due to the differing electron density around the phosphorus atom. As electronegativity increases, the halogen atoms pull electron density away from the phosphorus, affecting the spatial arrangement of the bonds.
Recommended video:
Guided course

Electronegativity Trends
Bond Angles
Bond angles are the angles formed between two bonds that share a common atom, which in this case is phosphorus in the trihalides. The bond angle can be influenced by factors such as lone pair repulsion and the size of the bonded atoms. In phosphorus trihalides, as the size and electronegativity of the halogen increase, the bond angles also change, reflecting the balance between repulsion and attraction in the molecular geometry.
Recommended video:
Guided course
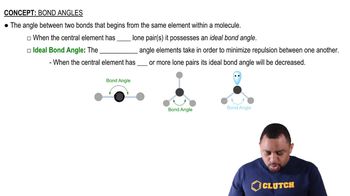
Bond Angles
Molecular Geometry
Molecular geometry refers to the three-dimensional arrangement of atoms within a molecule. The geometry of phosphorus trihalides is primarily determined by the number of bonding pairs and lone pairs of electrons around the phosphorus atom. The variations in bond angles observed in PF3, PCl3, PBr3, and PI3 can be attributed to the differences in steric effects and electronegativity of the halogens, which influence the overall shape and angles in the molecule.
Recommended video:
Guided course
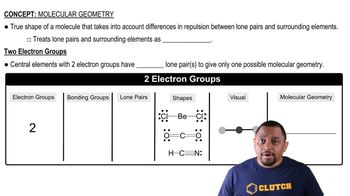
Molecular Geometry with Two Electron Groups
Related Practice
Textbook Question
400
views
Textbook Question
Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfur tetrafluoride monoxide (OSF4) according to the following unbalanced reaction: SF4(g) + O2(g) → OSF4(g) The O atom and the four F atoms in OSF4 are bonded to a central S atom. (c) Use average bond enthalpies (Table 8.3) to estimate the enthalpy of the reaction. Is it endothermic or exothermic?
688
views
Textbook Question
Sulfur tetrafluoride 1SF42 reacts slowly with O2 to form sulfur
tetrafluoride monoxide 1OSF42 according to the following
unbalanced reaction:
SF41g2 + O21g2¡OSF41g2
The O atom and the four F atoms in OSF4 are bonded to a
central S atom.
(e) For each of the molecules you drew in part (d), state how many
fluorines are equatorial and how many are axial.
102
views
Textbook Question
Many compounds of the transition-metal elements containdirect bonds between metal atoms. We will assumethat the z-axis is defined as the metal–metal bond axis.(d) Sketch the energyleveldiagram for the Sc2 molecule, assuming that only the3d orbital from part (a) is important.
353
views
