Textbook Question
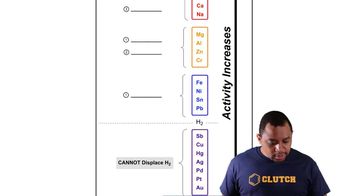
If Kc = 7.5×10−9 at 1000 K for the reaction N2(g) + O2(g) → 2 NO(g), give the value of Kc at 1000 K for the reaction
(a) 2 NO(g) → N2(g) + O2(g)
115
views

 Verified step by step guidance
Verified step by step guidance

If Kc = 7.5×10−9 at 1000 K for the reaction N2(g) + O2(g) → 2 NO(g), give the value of Kc at 1000 K for the reaction
(a) 2 NO(g) → N2(g) + O2(g)
If Kc = 7.5×10−9 at 1000 K for the reaction N2(g) + O2(g) → 2 NO(g), give the value of Kc at 1000 K for the reaction
(b) NO(g) → 1/2 N2(g) + 1/2 O2(g)