Textbook Question
Name an element in the fourth period (row) of the periodic table with the following: d. full s and p sublevels
1518
views

 Verified step by step guidance
Verified step by step guidance


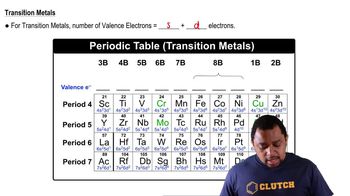
Name an element in the fourth period (row) of the periodic table with the following: d. full s and p sublevels
Name an element in the third period (row) of the periodic table with the following: a. three valence electrons
Name an element in the third period (row) of the periodic table with the following: b. four 3p electrons
Name an element in the third period (row) of the periodic table with the following: d. two 3s electrons and no 3p electrons
Determine the number of valence electrons in an atom of each element. a. Ba c. Ni d. S
Determine the number of valence electrons in an atom of each element. b. Cs