In the formate ion, HCO2-, the carbon atom is the central atom with the other three atoms attached to it. (d) How many electrons are in the p system of the ion?

d. Would you expect SO3 to exhibit delocalized 𝜋 bonding?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Delocalized π Bonding
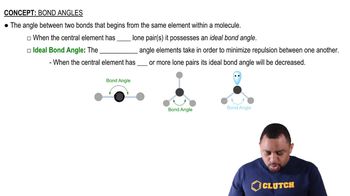
Resonance Structures

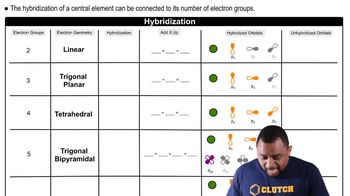
Molecular Geometry and Hybridization
In the formate ion, HCO2-, the carbon atom is the central atom with the other three atoms attached to it. (c) Are there multiple equivalent resonance structures for the ion?
Glycine, the simplest amino acid, has the following Lewis structure:
a. What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each of them?
Consider the following Lewis structure:
a. Does the Lewis structure depict a neutral molecule or an ion? If it is an ion, what is the charge on the ion?
Glycine, the simplest amino acid, has the following Lewis structure:
c. What is the total number of 𝜎 bonds in the entire molecule, and what is the total number of 𝜋 bonds?
Glycine, the simplest amino acid, has the following Lewis structure:
b. What are the hybridizations of the orbitals on the two oxygens and the nitrogen atom, and what are the approximate bond angles at the nitrogen?
