Textbook Question
Name each ionic compound. a. Mg3N2 b. KF c. Na2O d. Li2S f. KI e. CsF
775
views

 Verified step by step guidance
Verified step by step guidance

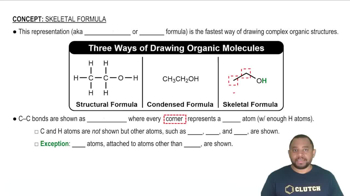

Name each ionic compound. a. Mg3N2 b. KF c. Na2O d. Li2S f. KI e. CsF
Classify each compound as ionic or molecular. a. CF2Cl2 b. CCl4 c. PtO2 d. SO3
Based on the molecular views, classify each substance as an atomic element, a molecular element, an ionic compound, or a molecular compound.
Name each ionic compound. a. SnCl4 b. PbI2 c. Fe2O3 d. CuI2 e. HgBr2 f. CrCl2
Write a formula for the compound that forms between potassium and each polyatomic ion. a. acetate b. phosphate c. sulfate d. nitrate
Based on the molecular views, classify each substance as an atomic element, a molecular element, an ionic compound, or a molecular compound.