Textbook Question
Identify each solid as molecular, ionic, or atomic. b. H2O(s)

 Verified step by step guidance
Verified step by step guidance
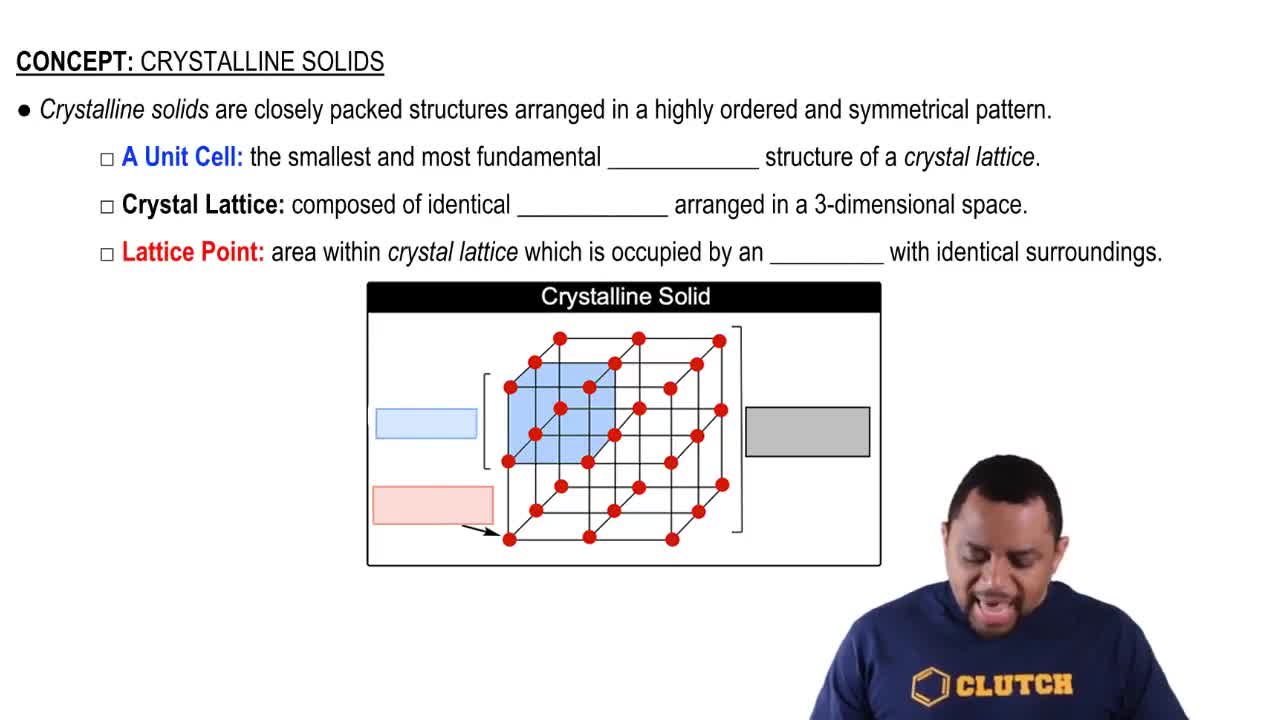

Identify each solid as molecular, ionic, or atomic. b. H2O(s)
Which solid has the highest melting point? Why? C(s, diamond), Kr(s), NaCl(s), H2O(s)
Identify each solid as molecular, ionic, or atomic. d. Xe(s)
Plonium crystallizes with a simple cubic structure. It has a density of 9.3 g/cm3, a radius of 167 pm, and a molar mass of 209 g/mol. Use these data to calculate Avogadro's number (the number of atoms in one mole).
Barium has a density of 3.59 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a barium atom.