The following diagram represents a buffer composed of equal concentrations of a weak acid, HA, and its conjugate base, A-. The heights of the columns are proportional to the concentrations of the components of the buffer. (c) Which of the three represents a situation that cannot arise from the addition of either an acid or a base? [Section 17.2]

A buffer contains a weak acid, HA, and its conjugate base. The weak acid has a pKa of 4.5, and the buffer has a pH of 4.3. Without doing a calculation, state which of these possibilities are correct at pH 4.3. (a) 3HA4 = 3A-4, (b) 3HA4 7 3A-4, or (c) 3HA4 6 3A-4. [Section 17.2]
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
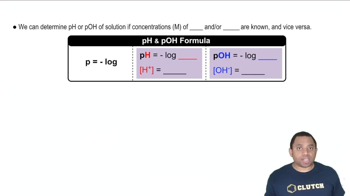
Buffer Solutions

Henderson-Hasselbalch Equation

pKa and pH Relationship
The following figure represents solutions at various stages of the titration of a weak acid, HA, with NaOH. (The Na+ ions and water molecules have been omitted for clarity.) To which of the following regions of the titration curve does each drawing correspond: (b) after addition of NaOH but before the equivalence point? [Section 17.3]
The beaker on the right contains 0.1 M acetic acid solution with methyl orange as an indicator. The beaker on the left contains a mixture of 0.1 M acetic acid and 0.1 M sodium acetate with methyl orange. (a) Using Figures 16.8 and 16.9, which solution has a higher pH?
The following diagram represents a buffer composed of equal concentrations of a weak acid, HA, and its conjugate base, A-. The heights of the columns are proportional to the concentrations of the components of the buffer. (a) Which of the three drawings, (1), (2), or (3), represents the buffer after the addition of a strong acid? [Section 17.2]
The beaker on the right contains 0.1 M acetic acid solution with methyl orange as an indicator. The beaker on the left contains a mixture of 0.1 M acetic acid and 0.1 M sodium acetate with methyl orange. (b) Which solution is better able to maintain its pH when small amounts of NaOH are added? Explain. [Sections 17.1 and 17.2]
