Consider the stable elements through lead (Z = 82). In how many instances are the atomic weights of the elements out of order relative to the atomic numbers of the elements?
Ch.7 - Periodic Properties of the Elements

Brown14th EditionChemistry: The Central ScienceISBN: 9780134414232Not the one you use?Change textbook
Chapter 7, Problem 80b
Figure 7.4 shows the radial probability distribution functions for the 2s orbitals and 2p orbitals. (b) How would you modify Slater's rules to adjust for the difference in electronic penetration of the nucleus for the 2s and 2p orbitals?
 Verified step by step guidance
Verified step by step guidance1
Step 1: Understand the concept of Slater's rules. Slater's rules are a set of empirical rules that estimate the effective nuclear charge, or the net positive charge experienced by an electron in a multi-electron atom. The rules take into account the shielding effect of other electrons, which reduces the net positive charge experienced by an electron.
Step 2: Understand the concept of electronic penetration. Electronic penetration refers to the ability of an electron to get close to the nucleus. In general, s electrons penetrate more effectively than p electrons, which means they experience a higher effective nuclear charge.
Step 3: Consider the difference between 2s and 2p orbitals. The 2s orbital is closer to the nucleus and more penetrating than the 2p orbital. Therefore, an electron in a 2s orbital will experience a higher effective nuclear charge than an electron in a 2p orbital.
Step 4: Modify Slater's rules. To adjust for the difference in electronic penetration of the nucleus for the 2s and 2p orbitals, you could assign a higher shielding constant to the 2p electrons than to the 2s electrons. This would reflect the fact that 2s electrons are more effective at penetrating the electron cloud and experiencing the nuclear charge.
Step 5: Apply the modified rules. Once you have adjusted Slater's rules, you can apply them to calculate the effective nuclear charge for electrons in different orbitals. Remember, the higher the effective nuclear charge, the more strongly an electron is attracted to the nucleus, and the lower its energy.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
2mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Radial Probability Distribution
The radial probability distribution function describes the likelihood of finding an electron at a certain distance from the nucleus in an atom. It is derived from the square of the wave function and provides insight into the spatial distribution of electrons in different orbitals, such as 2s and 2p. Understanding this concept is crucial for analyzing how electron density varies with distance and how it influences chemical behavior.
Recommended video:
Guided course

Velocity Distributions
Slater's Rules
Slater's rules are a set of guidelines used to estimate the effective nuclear charge experienced by an electron in a multi-electron atom. These rules take into account the shielding effect of other electrons, which can vary based on their orbital types. Modifying Slater's rules for different orbitals, like 2s and 2p, involves considering their differing penetration abilities and how they shield each other from the nucleus.
Recommended video:
Guided course

Effective Nuclear Charge Calculation with Slater's Rules
Electron Penetration
Electron penetration refers to the ability of an electron in a given orbital to get close to the nucleus, which affects its energy and the effective nuclear charge it experiences. The 2s orbital has a higher penetration compared to the 2p orbital, meaning that 2s electrons can shield the nucleus more effectively. This difference is significant when applying Slater's rules, as it influences the calculated effective nuclear charge for electrons in these orbitals.
Recommended video:
Guided course
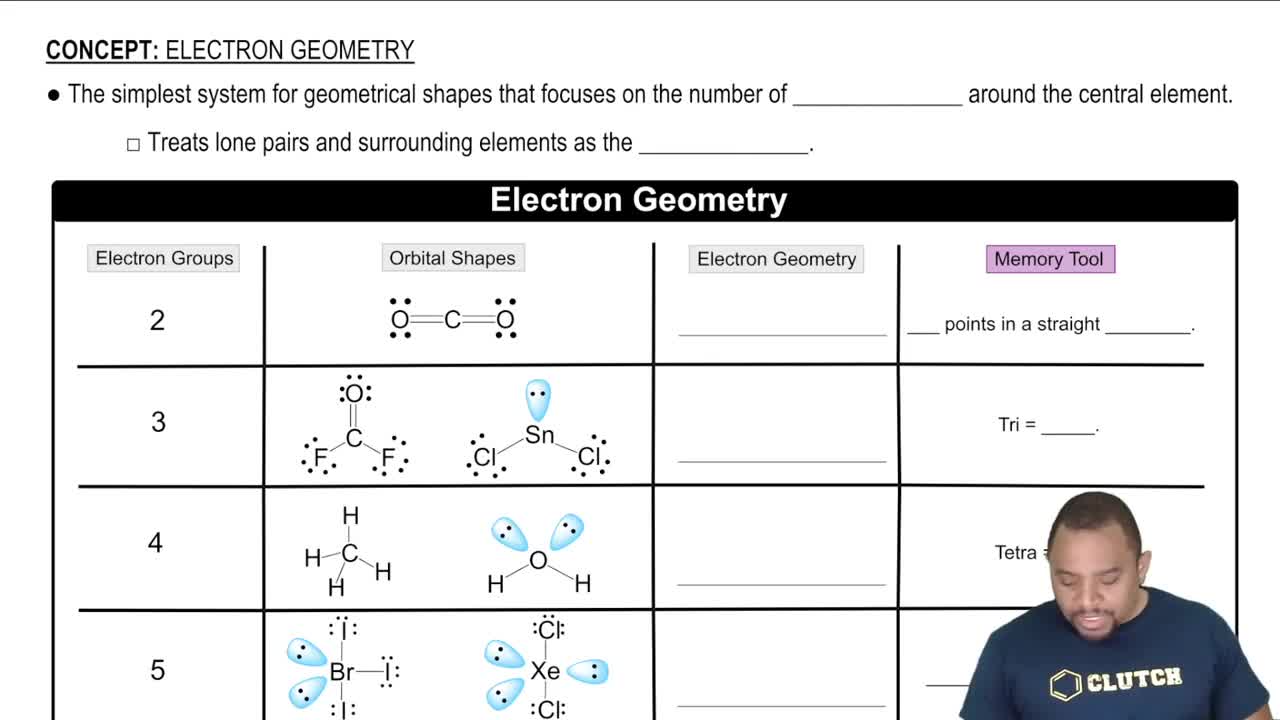
Electron Geometry
Related Practice
Textbook Question
483
views
Textbook Question
Figure 7.4 shows the radial probability distribution functions for the 2s orbitals and 2p orbitals. (a) Which orbital, 2s or 2p, has more electron density close to the nucleus?
843
views
Textbook Question
(a) If the core electrons were totally effective at screening the valence electrons and the valence electrons provided no screening for each other, what would be the effective nuclear charge acting on the 3s and 3p valence electrons in P?
1082
views
Textbook Question
(b) Repeat these calculations using Slater’s rules.
996
views
Textbook Question
(c) Detailed calculations indicate that the effective nuclear charge is 5.6+ for the 3s electrons and 4.9+ for the 3p electrons. Why are the values for the 3s and 3p electrons different?
