Which of the following schematic drawings best describes a solution of Li2SO4 in water (water molecules not shown for simplicity)?

The concept of chemical equilibrium is very important. Which one of the following statements is the most correct way to think about equilibrium? (a) If a system is at equilibrium, nothing is happening. (b) If a system is at equilibrium, the rate of the forward reaction is equal to the rate of the back reaction. (c) If a system is at equilibrium, the product concentration is changing over time.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
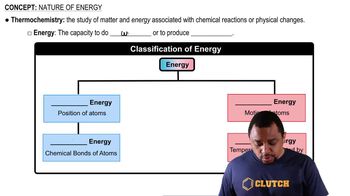
Key Concepts
Chemical Equilibrium

Dynamic Nature of Equilibrium
Equilibrium Constant

Aqueous solutions of three different substances, AX, AY, and AZ, are represented by the three accompanying diagrams. Identify each substance as a strong electrolyte, a weak electrolyte, or a nonelectrolyte.
Use the molecular representations shown here to classify each compound as a nonelectrolyte, a weak electrolyte, or a strong electrolyte (see Figure 4.6 for the element color scheme). (a)
You are presented with a white solid and told that due to careless labeling it is not clear if the substance is barium chloride, lead chloride, or zinc chloride. When you transfer the solid to a beaker and add water, the solid dissolves to give a clear solution. Next an Na2SO41aq2 solution is added and a white precipitate forms. What is the identity of the unknown white solid?
Which of the following ions will always be a spectator ion in a precipitation reaction? (a) Cl- (b) NO3- (c) NH4+ (d) S2- (e) SO42-
