(b) Is this line in the visible region of the electromagnetic spectrum?

The visible emission lines observed by Balmer all involved nf = 2. (b) Calculate the wavelengths of the first three lines in the Balmer series—those for which ni = 3, 4, and 5—and identify these lines in the emission spectrum shown in Figure 6.11.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Balmer Series

Rydberg Formula
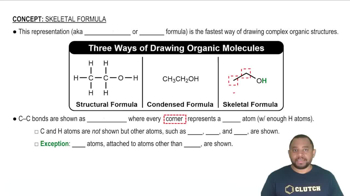
Emission Spectrum

Consider a transition of the electron in the hydrogen atom from n = 8 to n = 3. (b) Will the light be absorbed or emitted?
The visible emission lines observed by Balmer all involved nf = 2. (a) Which of the following is the best explanation of why the lines with nf = 3 are not observed in the visible portion of the spectrum: (i) Transitions to nf = 3 are not allowed to happen, (ii) transitions to nf = 3 emit photons in the infrared portion of the spectrum, (iii) transitions to nf = 3 emit photons in the ultraviolet portion of the spectrum, or (iv) transitions to nf = 3 emit photons that are at exactly the same wavelengths as those to nf = 2.
The Lyman series of emission lines of the hydrogen atom are those for which nf = 1. (a) Determine the region of the electromagnetic spectrum in which the lines of the Lyman series are observed.
The Lyman series of emission lines of the hydrogen atom are those for which nf = 1. (b) Calculate the wavelengths of the first three lines in the Lyman series—those for which ni = 2, 3, and 4.
One of the emission lines of the hydrogen atom has a wavelength of 94.974 nm. (a) In what region of the electromagnetic spectrum is this emission found?
