Write orbital diagrams (boxes with arrows in them) to represent the electron configurations of carbon before and after sp hybridization.

 Tro 4th Edition
Tro 4th Edition Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory
Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory Problem 61a
Problem 61aWrite a hybridization and bonding scheme for each molecule. Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 10.6 and 10.7. a. CCl4
 Verified step by step guidance
Verified step by step guidance
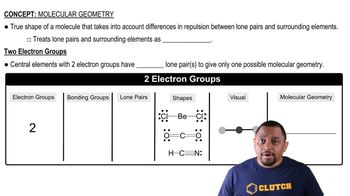
Verified video answer for a similar problem:
Key Concepts
Hybridization

Bonding and Molecular Geometry
Orbital Overlap Theory

Which hybridization scheme allows the formation of at least one p bond? sp3, sp2, sp3d2
Which hybridization scheme allows the central atom to form more than four bonds? sp3, sp3d, sp2
Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 10.6 and 10.7. b. NH3
Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 10.6 and 10.7. c. OF2 d. CO2
Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 10.6 and 10.7.a. CH2Br2 b. SO2 c. NF3 d. BF3