Match each of the Lewis structures (a to c) with the correct diagram (1 to 3) of its shape, and name the shape; indicate if each molecule is polar or nonpolar. Assume X and Y are nonmetals and all bonds are polar covalent.
<IMAGE>
c.

 Verified step by step guidance
Verified step by step guidance
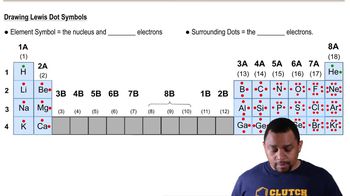
Match each of the Lewis structures (a to c) with the correct diagram (1 to 3) of its shape, and name the shape; indicate if each molecule is polar or nonpolar. Assume X and Y are nonmetals and all bonds are polar covalent.
<IMAGE>
c.
Consider an ion with the symbol X2+ formed from a representative element.
d. What is the formula of the compound formed from X and the nitride ion?
Consider the following bonds: Ca and O, C and O, K and O, O and O, and N and O.
d. Arrange the covalent bonds in order of decreasing polarity.
Consider an ion with the symbol X2+ formed from a representative element.
c. If X is in Period 3, what is the element?
Consider an ion with the symbol Y3- formed from a representative element.
b. What is the Lewis symbol of the element?
Match each of the formulas (a to c) with the correct diagram (1 to 3) of its shape, and name the shape; indicate if each molecule is polar or nonpolar.
<IMAGE>
a. PBr3