
Where learning comes to life
Explore learning for every stage of life with Pearson.
For School
Inspire a lifelong love of learning and build future-ready foundations.
For College
Deliver practical, hands-on learning that turns passion into profession.
For University
Support deep academic learning with practical, future-ready outcomes.
For Work
Attract and upskill the workforce for today’s needs and tomorrow’s ambitions.
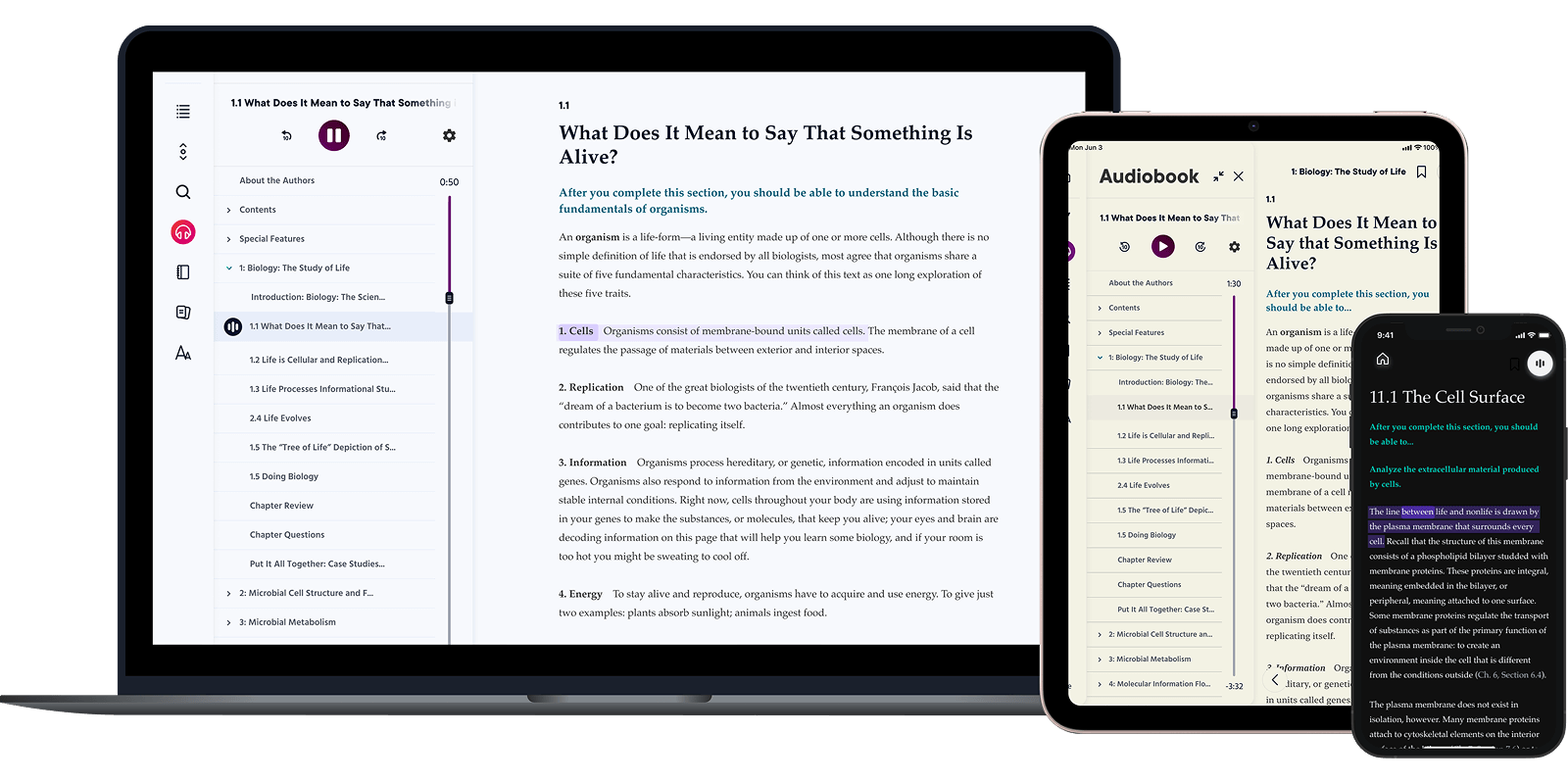
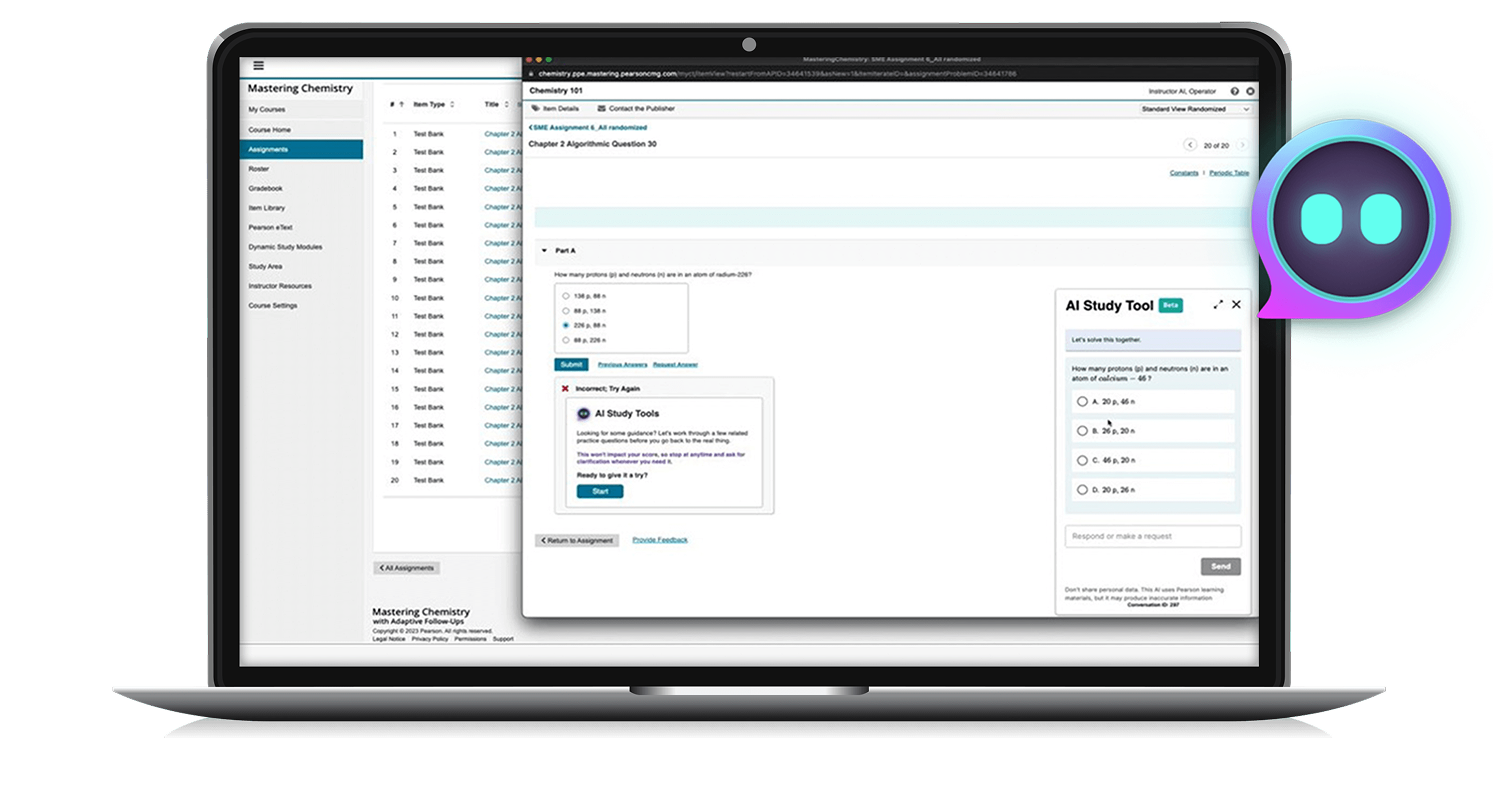
Learning never stops
The more we learn, the more we achieve.

About Pearson
We create meaningful learning experiences for people of all ages. Our mission? To help students, educators, and businesses make progress toward their personal and professional goals.
Purpose driven
The right learning solutions make a big difference at every age. We help people realise the lives they imagine through learning.
Digital first
Flexible, affordable tech solutions fit the way you live and learn. That's why we focus on adaptability and innovation.
