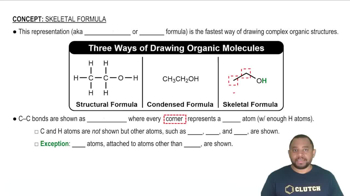
Textbook Question
Little is known about the properties of astatine, At, because of its rarity and high radioactivity. Nevertheless, it is possible for us to make many predictions about its properties. (a) Do you expect the element to be a gas, liquid, or solid at room temperature?
989
views