Textbook Question
A molecule with the formula AB3 has a trigonal pyramidal geometry. How many electron groups are on the central atom (A)?
3053
views

 Tro 5th Edition
Tro 5th Edition Ch.11 - Chemical Bonding II: Molecular Shapes, VSEPR & MO Theory
Ch.11 - Chemical Bonding II: Molecular Shapes, VSEPR & MO Theory Problem 35
Problem 35 Verified step by step guidance
Verified step by step guidance
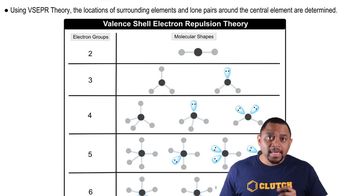
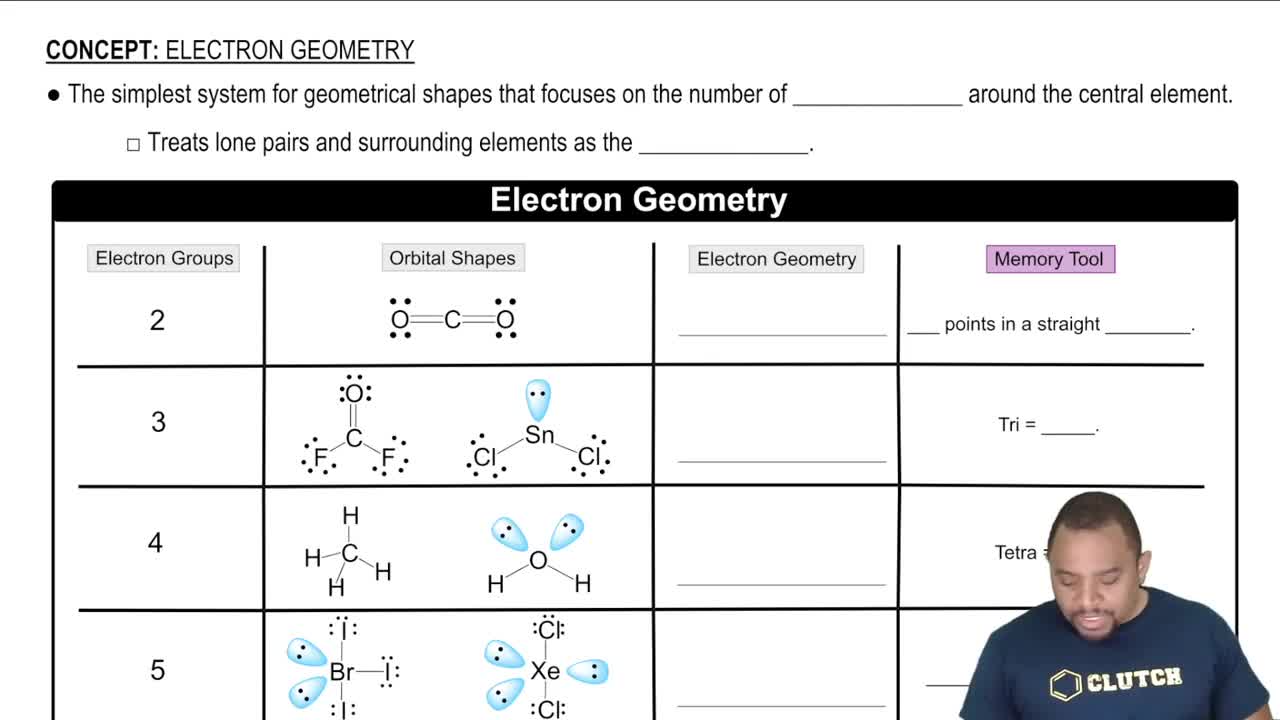
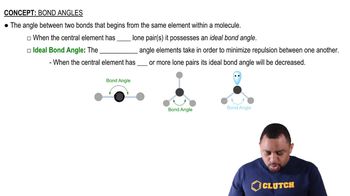
A molecule with the formula AB3 has a trigonal pyramidal geometry. How many electron groups are on the central atom (A)?
A molecule with the formula AB3 has a trigonal planar geometry. How many electron groups are on the central atom?
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom. (c)
Determine the electron geometry, molecular geometry, and idealized bond angles for each molecule. In which cases do you expect deviations from the idealized bond angle? a. CF4 b. NF3 c. OF2 d. H2S
Which species has the smaller bond angle, H3O+ or H2O? Explain.
Which species has the smaller bond angle, ClO4- or ClO3- ? Explain.