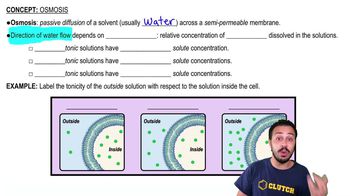
If a solution surrounding a cell is hypertonic relative to the inside of the cell, how will water move?
a. It will move into the cell via osmosis.
b. It will move out of the cell via osmosis.
c. It will not move, because equilibrium exists.
d. It will evaporate from the cell surface more rapidly.