In the study of chemistry, dimensional analysis plays a crucial role in solving complex problems that involve unit conversions. At the heart of dimensional analysis is the concept of the conversion factor, which is a ratio that relates two different units. For instance, the relationship between days and hours can be expressed as a conversion factor: one day equals 24 hours. This can be represented in fractional form as either:
\[ \text{1 day} = \frac{24 \text{ hours}}{1} \quad \text{or} \quad \text{24 hours} = \frac{1 \text{ day}}{1} \]
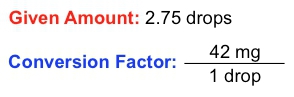
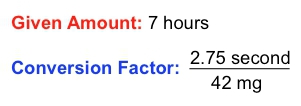
This demonstrates how different units can be interconnected through conversion factors. Another important concept is the given amount, which refers to a value expressed in a single unit without any conversion. For example, if you study for 3 hours, the given amount is simply 3 hours, without any relation to other units.
Understanding how to identify and utilize conversion factors and given amounts is essential for mastering dimensional analysis. This skill will enable you to tackle various problems effectively, especially as you prepare for quizzes and exams in chemistry. As you progress, you will encounter exercises that require you to pinpoint these elements within different questions, enhancing your analytical abilities in the subject.