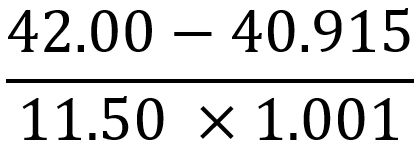In chemistry, understanding significant figures is crucial for accurate calculations, especially when performing multiplication and division. The key principle to remember is that the final answer should reflect the least number of significant figures from the values used in the calculation.
For example, consider the multiplication of three values: 3.16, 0.003027, and \(5.7 \times 10^{-3}\). To determine the number of significant figures in each value, we start counting from the first non-zero digit:
- For 3.16, the first non-zero digit is 3, and counting from there gives us 3 significant figures.
- For 0.003027, the first non-zero digit is 3, leading to 4 significant figures.
- For \(5.7 \times 10^{-3}\), the coefficient is 5, which gives us 2 significant figures.
Among these, the value with the least significant figures is \(5.7 \times 10^{-3}\) with 2 significant figures. Therefore, the final answer must also be expressed with 2 significant figures.
After performing the multiplication, the initial result is \(5.4522324 \times 10^{-5}\). To round this to 2 significant figures, we look at the first two digits, 5 and 4. The next digit is 5, which means we round up the 4 to 5. Thus, the final answer is \(5.5 \times 10^{-5}\), ensuring it adheres to the significant figures rule based on the original values.
Having covered multiplication and division, it is essential to explore how significant figures apply to addition and subtraction in subsequent calculations.